
- •Contents
- •Contributors
- •Brain Tumor Imaging
- •1 Introduction
- •1.1 Overview
- •2 Clinical Management
- •3 Glial Tumors
- •3.1 Focal Glial and Glioneuronal Tumors Versus Diffuse Gliomas
- •3.3 Astrocytomas Versus Oligodendroglial Tumors
- •3.4.1 Diffuse Astrocytoma (WHO Grade II)
- •3.5 Anaplastic Glioma (WHO Grade III)
- •3.5.1 Anaplastic Astrocytoma (WHO Grade III)
- •3.5.3 Gliomatosis Cerebri
- •3.6 Glioblastoma (WHO Grade IV)
- •4 Primary CNS Lymphomas
- •5 Metastatic Tumors of the CNS
- •References
- •MR Imaging of Brain Tumors
- •1 Introduction
- •2 Brain Tumors in Adults
- •2.1 Questions to the Radiologist
- •2.2 Tumor Localization
- •2.3 Tumor Malignancy
- •2.4 Tumor Monitoring
- •2.5 Imaging Protocol
- •Computer Tomography
- •2.6 Case Illustrations
- •3 Pediatric Brain Tumors
- •3.1 Standard MRI
- •3.2 Differential Diagnosis of Common Pediatric Brain Tumors
- •3.3 Early Postoperative Imaging
- •3.4 Meningeal Dissemination
- •References
- •MR Spectroscopic Imaging
- •1 Methods
- •1.1 Introduction to MRS
- •1.2 Summary of Spectroscopic Imaging Techniques Applied in Tumor Diagnostics
- •1.3 Partial Volume Effects Due to Low Resolution
- •1.4 Evaluation of Metabolite Concentrations
- •1.5 Artifacts in Metabolite Maps
- •2 Tumor Metabolism
- •3 Tumor Grading and Heterogeneity
- •3.1 Some Aspects of Differential Diagnosis
- •4 Prognostic Markers
- •5 Treatment Monitoring
- •References
- •MR Perfusion Imaging
- •1 Key Points
- •2 Methods
- •2.1 Exogenous Tracer Methods
- •2.1.1 Dynamic Susceptibility Contrast MRI
- •2.1.2 Dynamic Contrast-Enhanced MRI
- •3 Clinical Application
- •3.1 General Aspects
- •3.3 Differential Diagnosis of Tumors
- •3.4 Tumor Grading and Prognosis
- •3.5 Guidance for Biopsy and Radiation Therapy Planning
- •3.6 Treatment Monitoring
- •References
- •Diffusion-Weighted Methods
- •1 Methods
- •2 Microstructural Changes
- •4 Prognostic Marker
- •5 Treatment Monitoring
- •Conclusion
- •References
- •1 MR Relaxometry Techniques
- •2 Transverse Relaxation Time T2
- •4 Longitudinal Relaxation Time T1
- •6 Cest Method
- •7 CEST Imaging in Brain Tumors
- •References
- •PET Imaging of Brain Tumors
- •1 Introduction
- •2 Methods
- •2.1 18F-2-Fluoro-2-Deoxy-d-Glucose
- •2.2 Radiolabeled Amino Acids
- •2.3 Radiolabeled Nucleoside Analogs
- •2.4 Imaging of Hypoxia
- •2.5 Imaging Angiogenesis
- •2.6 Somatostatin Receptors
- •2.7 Radiolabeled Choline
- •3 Delineation of Tumor Extent, Biopsy Guidance, and Treatment Planning
- •4 Tumor Grading and Prognosis
- •5 Treatment Monitoring
- •7 PET in Patients with Brain Metastasis
- •8 Imaging of Brain Tumors in Children
- •9 Perspectives
- •References
- •1 Treatment of Gliomas and Radiation Therapy Techniques
- •2 Modern Methods and Strategies
- •2.2 3D Conformal Radiation Therapy
- •2.4 Stereotactic Radiosurgery (SRS) and Radiotherapy
- •2.5 Interstitial Brachytherapy
- •2.6 Dose Prescription
- •2.7 Particle Radiation Therapy
- •3 Role of Imaging and Treatment Planning
- •3.1 Computed Tomography (CT)
- •3.2 Magnetic Resonance Imaging (MRI)
- •3.3 Positron Emission Tomography (PET)
- •4 Prognosis
- •Conclusion
- •References
- •1 Why Is Advanced Imaging Indispensable for Modern Glioma Surgery?
- •2 Preoperative Imaging Strategies
- •2.4 Preoperative Imaging of Function and Functional Anatomy
- •2.4.1 Imaging of Functional Cortex
- •2.4.2 Imaging of Subcortical Tracts
- •3 Intraoperative Allocation of Relevant Anatomy
- •Conclusions
- •References
- •Future Methods in Tumor Imaging
- •1 Special Editing Methods in 1H MRS
- •1.1 Measuring Glycine
- •2 Other Nuclei
- •2.1.1 Spatial Resolution
- •2.1.2 Measuring pH
- •2.1.3 Measuring Lipid Metabolism
- •2.1.4 Energy Metabolism
- •References

Future Methods in Tumor Imaging
Ulrich Pilatus and Elke Hattingen
Contents |
|
Abstract |
|
|
1 Special Editing Methods in 1H MRS |
155 |
Chapter Advanced MR Methods in Differential Diagnosis |
||
1.1 |
Measuring Glycine |
156 |
of Brain Tumors deals with advanced and future MR |
|
1.2 |
Measuring 2-hydroxyglutarate |
156 |
imaging methods in brain tumors. In this chapter, we will |
|
2 Other Nuclei |
157 |
discuss future MR spectroscopic methods that are promis- |
||
2.1 |
31P MRS |
157 |
ing regarding the tumor diagnosis and the research of |
|
2.2 |
13C MRS |
159 |
tumor biology. Whereas Chap. MR Spectroscopic |
|
References |
160 |
Imaging focuses on diagnostic signiÞcance of 1H and 31P |
||
|
|
|
MRS, we herein put more emphasis on methodical issues |
|
|
|
|
of MRS. First, we deal with special editing methods to |
|
|
|
|
detect special ÒtumorÓ metabolites (glycine, 2-hydroxy- |
|
|
|
|
glutarate). In the second part, we discuss methods and |
|
biological implications of x-nucleus spectroscopy, focusing on the nuclei 31P and 13C. Here, we point out that a considerable proportion of advanced spectroscopic studies dealing with brain tumors come from animal studies.
U. Pilatus (*)
Department of Neuroradiology, Goethe University Frankfurt, Schleusenweg 2-16, Frankfurt 60528, Germany
e-mail: u.pilatus@em.uni-frankfurt.de
E. Hattingen
Neuroradiology, Clinic of Rheinische Friedrich-Wilhelms- University, Sigmund-Freud Stra§e 6, 53127 Bonn, Germany e-mail: elke.hattingen@ukb.uni-bonn.de
E. Hattingen, U. Pilatus (eds.), Brain Tumor Imaging, Med Radiol Diagn Imaging (2016),
DOI 10.1007/174_2016_1055, © Springer Berlin Heidelberg
Abbreviations
2-HG |
2-hydroxyglutarate |
ATP |
Adenosine triphosphate |
Gly |
Glycine |
GPC |
Glycerophosphocholine |
MI |
Myo-inositol |
PCho |
Phosphocholine |
PCr |
Phosphocreatine |
Pi |
Inorganic phosphate |
TCA cycle |
Tricarboxylic acid cycle or Krebs cycle |
tCho |
Total choline |
tCr |
Total creatine |
tNAA |
Total N-acetylaspartate |
1Special Editing Methods in 1H MRS
In Chap. MR Spectroscopic Imaging, spectroscopic methods were described which provide biochemical information. In addition to the easily detectable main metabolites (creatine,
155
156 |
U. Pilatus and E. Hattingen |
|
|
choline, tNAA), other compounds can be detected using more sophisticated technique markers, which may serve as biomarkers for speciÞc questions in tumor diagnosis. These are glycine and 2-hydroxyglutarate (2-HG), which require nonstandard techniques like speciÞc spacing of the refocusing pulses in PRESS or additional RF pulses in the sequence like MEGA PRESS.
1.1Measuring Glycine
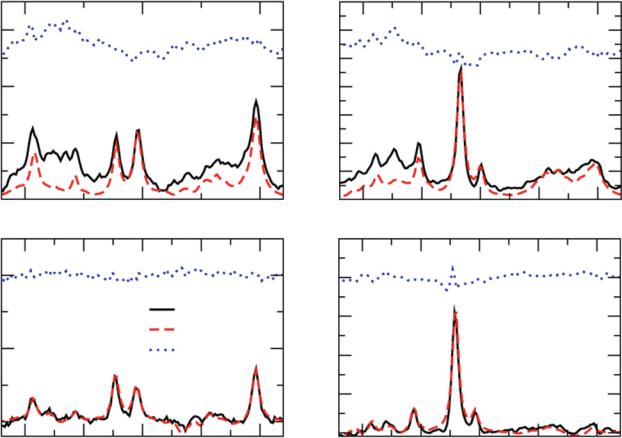
At short echo time (TE) of about 30 ms, the glycine (Gly) signal, a singlet at 3.56 ppm, is masked by the main peak of myo-inositol (MI). Since MI represents a strongly coupled spin system (Govindaraju et al. 1998), its pattern is signiÞcantly changing with TE, exhibiting a signal reduction at TE of 135Ð44, which is far beyond the signal decay due to typical T2 relaxation of singlets. This effect can be exploited for differentiation between MI and Gly as has been shown in several studies on brain tumors using either both TE
(Hattingen et al. 2009; Davies et al. 2010) or performing a single spectroscopic measurement with an optimized TE and refocusing pulse spacing in addition to dedicated spectral analysis (Choi et al. 2011; Maudsley et al. 2014). Figure 1 shows the efÞciency of using the long and short TE, comparing tumor spectra for TE at 30 ms to those obtained at 144 ms. It is obvious that there is no signiÞcant decrease in the 3.56 ppm signal between the tumor spectra (b, d). In contrast, rather small signal at TE of 144 ms in normal-appearing tissue (c) indicates the rather low concentration of MI here.
1.2Measuring 2-hydroxyglutarate
About 70 % of WHO grade II and III gliomas have a mutation of isocitrate dehydrogenase (IDH1 and IDH2). These mutations cause an increased formation of 2-hydroxyglutarate (2-HG) from isocitrate, offering the obvious approach to consider increased 2-HG as a tumor marker visible in vivo
a |
|
|
|
b |
7 |
|
|
|
|
3 |
|
|
|
|
6 |
|
Cho |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
5 |
|
|
|
|
2 |
|
|
|
NAA |
4 |
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
Cho tCr |
|
|
3 |
MI/Gly |
|
|
|
|
MI/Gly |
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
1 |
|
|
|
|
2 |
|
tCr |
|
|
|
|
|
|
|
1 |
|
|
|
|
0 |
|
|
|
|
0 |
|
|
|
|
4 |
|
3 |
|
2 |
4 |
3.5 |
3 |
2.5 |
2 |
c |
|
|
|
d |
5 |
|
|
|
|
2 |
|
|
|
|
4 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Raw data |
|
3 |
|
|
|
|
|
|
|
jMRU fit |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1 |
|
|
Residue |
|
2 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1 |
|
|
|
|
0 |
|
|
|
|
0 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4 |
3.5 |
3 |
2.5 |
2 |
4 |
3.5 |
3 |
2.5 |
2 |
|
|
Frequency (ppm) |
|
|
|
|
Frequency (ppm) |
|
|
Fig. 1 Discriminating glycine from myo-inositol by comparing short and long TE spectra. a shows a spectrum from normal tissue and b from tumor tissue at TE of 30 ms. The signal at 3.56 ppm could be Gly or MI. c shows the normal tissue at a TE of 144 ms, while d shows the repec-
tive tumor voxel. Normal tissue is known to contain MI but rather low concentrations of glycine. The lack of the 3.56 signal in c indicates the sufÞcient suppression of MI at long TE. (Black raw data, red signal estimated by data analyisis, blue residual)
