
- •Contents
- •Contributors
- •Brain Tumor Imaging
- •1 Introduction
- •1.1 Overview
- •2 Clinical Management
- •3 Glial Tumors
- •3.1 Focal Glial and Glioneuronal Tumors Versus Diffuse Gliomas
- •3.3 Astrocytomas Versus Oligodendroglial Tumors
- •3.4.1 Diffuse Astrocytoma (WHO Grade II)
- •3.5 Anaplastic Glioma (WHO Grade III)
- •3.5.1 Anaplastic Astrocytoma (WHO Grade III)
- •3.5.3 Gliomatosis Cerebri
- •3.6 Glioblastoma (WHO Grade IV)
- •4 Primary CNS Lymphomas
- •5 Metastatic Tumors of the CNS
- •References
- •MR Imaging of Brain Tumors
- •1 Introduction
- •2 Brain Tumors in Adults
- •2.1 Questions to the Radiologist
- •2.2 Tumor Localization
- •2.3 Tumor Malignancy
- •2.4 Tumor Monitoring
- •2.5 Imaging Protocol
- •Computer Tomography
- •2.6 Case Illustrations
- •3 Pediatric Brain Tumors
- •3.1 Standard MRI
- •3.2 Differential Diagnosis of Common Pediatric Brain Tumors
- •3.3 Early Postoperative Imaging
- •3.4 Meningeal Dissemination
- •References
- •MR Spectroscopic Imaging
- •1 Methods
- •1.1 Introduction to MRS
- •1.2 Summary of Spectroscopic Imaging Techniques Applied in Tumor Diagnostics
- •1.3 Partial Volume Effects Due to Low Resolution
- •1.4 Evaluation of Metabolite Concentrations
- •1.5 Artifacts in Metabolite Maps
- •2 Tumor Metabolism
- •3 Tumor Grading and Heterogeneity
- •3.1 Some Aspects of Differential Diagnosis
- •4 Prognostic Markers
- •5 Treatment Monitoring
- •References
- •MR Perfusion Imaging
- •1 Key Points
- •2 Methods
- •2.1 Exogenous Tracer Methods
- •2.1.1 Dynamic Susceptibility Contrast MRI
- •2.1.2 Dynamic Contrast-Enhanced MRI
- •3 Clinical Application
- •3.1 General Aspects
- •3.3 Differential Diagnosis of Tumors
- •3.4 Tumor Grading and Prognosis
- •3.5 Guidance for Biopsy and Radiation Therapy Planning
- •3.6 Treatment Monitoring
- •References
- •Diffusion-Weighted Methods
- •1 Methods
- •2 Microstructural Changes
- •4 Prognostic Marker
- •5 Treatment Monitoring
- •Conclusion
- •References
- •1 MR Relaxometry Techniques
- •2 Transverse Relaxation Time T2
- •4 Longitudinal Relaxation Time T1
- •6 Cest Method
- •7 CEST Imaging in Brain Tumors
- •References
- •PET Imaging of Brain Tumors
- •1 Introduction
- •2 Methods
- •2.1 18F-2-Fluoro-2-Deoxy-d-Glucose
- •2.2 Radiolabeled Amino Acids
- •2.3 Radiolabeled Nucleoside Analogs
- •2.4 Imaging of Hypoxia
- •2.5 Imaging Angiogenesis
- •2.6 Somatostatin Receptors
- •2.7 Radiolabeled Choline
- •3 Delineation of Tumor Extent, Biopsy Guidance, and Treatment Planning
- •4 Tumor Grading and Prognosis
- •5 Treatment Monitoring
- •7 PET in Patients with Brain Metastasis
- •8 Imaging of Brain Tumors in Children
- •9 Perspectives
- •References
- •1 Treatment of Gliomas and Radiation Therapy Techniques
- •2 Modern Methods and Strategies
- •2.2 3D Conformal Radiation Therapy
- •2.4 Stereotactic Radiosurgery (SRS) and Radiotherapy
- •2.5 Interstitial Brachytherapy
- •2.6 Dose Prescription
- •2.7 Particle Radiation Therapy
- •3 Role of Imaging and Treatment Planning
- •3.1 Computed Tomography (CT)
- •3.2 Magnetic Resonance Imaging (MRI)
- •3.3 Positron Emission Tomography (PET)
- •4 Prognosis
- •Conclusion
- •References
- •1 Why Is Advanced Imaging Indispensable for Modern Glioma Surgery?
- •2 Preoperative Imaging Strategies
- •2.4 Preoperative Imaging of Function and Functional Anatomy
- •2.4.1 Imaging of Functional Cortex
- •2.4.2 Imaging of Subcortical Tracts
- •3 Intraoperative Allocation of Relevant Anatomy
- •Conclusions
- •References
- •Future Methods in Tumor Imaging
- •1 Special Editing Methods in 1H MRS
- •1.1 Measuring Glycine
- •2 Other Nuclei
- •2.1.1 Spatial Resolution
- •2.1.2 Measuring pH
- •2.1.3 Measuring Lipid Metabolism
- •2.1.4 Energy Metabolism
- •References
34 |
E. Hattingen and M. Warmuth-Metz |
|
|
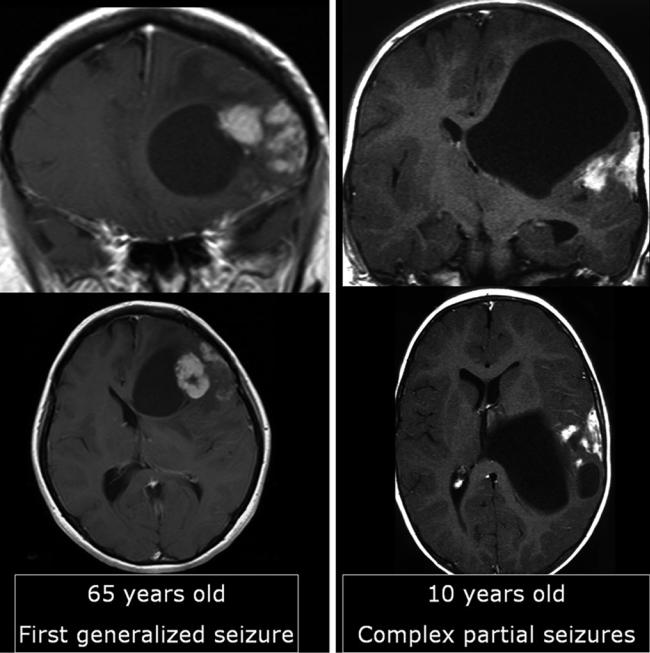
Fig. 29 Two similar tumors with enhancing nodules abutting the brain surface and large cysts: The tumor of the older patient shows some edema, whereas the tumor of the young patient lacks any edema. The
older patient only had one seizure, whereas the young patient had epilepsy with complex partial seizures. The older patient had a GBM; the young patient had a pleomorphic xanthoastrocytoma
show more or less pronounced calcification on CCT. Further, it should be performed in patients with MRI contraindications. CCT may also be helpful to characterize cell density of a brain tumor in children (Sect. 3). In adults PCNSL are typical tumors with cortex-isodense appearance in contrast to most of the gliomas which are hypodense on CCT.
2.6Case Illustrations
Although MRI has limited diagnostic accuracy, there are some clues to narrow differential diagnosis. These criteria should always include or rather begin with patient’s age considering that the likelihood of malignancy in adulthood increases with age (Fig. 29).

MR Imaging of Brain Tumors |
35 |
|
|
Fig. 30 Two similar histopathologically proven astrocytomas in the right parietal lobe infiltrating the cortex, both without contrast enhancement: The gyri are bloated and gray-white matter junction is effaced.
The tumor in (a) was a astrocytoma WHO grade III, whereas in (b) an astrocytoma WHO grade II was found
36 |
E. Hattingen and M. Warmuth-Metz |
|
|
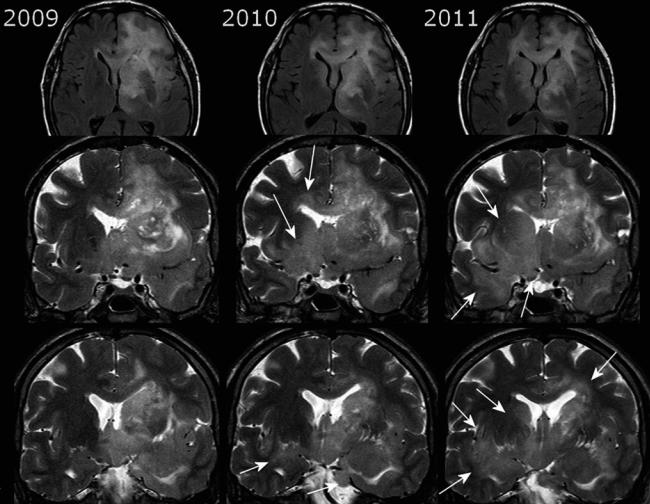
Fig. 31 The gliomatosis of the left hemisphere infiltrating the corpus callosum was treated with radiation in 2009. In 2010, the mass effect of the left-sided tumor declined, but tumor progressed by infiltrating the right temporal white matter, the insula, the basal ganglia, and
hypothalamus bilaterally (white arrows). Coronal T2WI depicts the infiltration ways en face minimizing partial volume effects of axial slices, and further they visualize the blurring of gray-white matter junction due to the tumor infiltration
1.WHO grade II and III gliomas
Most low-grade gliomas or gliomas with secondary
malignant transformation infiltrate cortical structures (Fig. 30). A preferential localization of low-grade astrocytoma is the frontotemporal lobe with involvement of the temporal stem, the temporomesial structures, and the insula. The infiltration way often follows white matter tracts like the anterior commissure and the corpus callosum. Coronaloriented T2WI are very helpful in depicting the extent of low-grade tumorsand to detect tumor progression in the follow-up (Fig. 31). Due to the diffuse infiltration pattern, low-grade tumors do not expand spherically, but infiltrate the anatomical structures often without severally
distorting them. Therefore, size measurement of these brain tumors is of limited value.
WHO grade III oligodendrogliomas are i.a. defined by microvascular proliferation and necrosis which give them the appearance of a highly malignant GBM (Fig. 32). WHO grade II oligodendrogliomas may already appear malignant due to their inhomogeneous tumor areas with cysts, calcifications, and hemorrhage (Fig. 33).
2.Glioneuronal tumors
The hallmarks of these tumors are the younger patient age
(children and young adults) and the high association with epilepsy with complex partial seizures. Although mostly benign, one should be aware that the glial component of

MR Imaging of Brain Tumors |
37 |
|
|
Fig. 32 Very inhomogeneous tumor with cystic areas, irregular, and patchy enhancement of the margins. It has the appearance of GBM, except for the small surrounding edema which is not typical for large
necrotic GBMs (a, b). The CCT (a) shows calcification which yields faint hyperand hypointense signal changes in precontrast T1WI (c); see also Fig. 33. Histopathology yielded an oligodendroglioma WHO grade III
