
- •Foreword
- •Preface
- •Contents
- •About the Editors
- •Contributors
- •1: Tracheobronchial Anatomy
- •Trachea
- •Introduction
- •External Morphology
- •Internal Morphology
- •Mucous Layer
- •Blood Supply
- •Anatomo-Clinical Relationships
- •Bronchi
- •Main Bronchi
- •Bronchial Division
- •Left Main Bronchus (LMB)
- •Right Main Bronchus (RMB)
- •Blood Supply
- •References
- •2: Flexible Bronchoscopy
- •Introduction
- •History
- •Description
- •Indications and Contraindications
- •Absolute Contraindications
- •Procedure Preparation
- •Technique of FB Procedure
- •Complications of FB Procedure
- •Basic Diagnostic Procedures
- •Bronchoalveolar Lavage (BAL)
- •Transbronchial Lung Biopsy (TBLB)
- •Transbronchial Needle Aspiration (TBNA)
- •Bronchial Brushings
- •Advanced Diagnostic Bronchoscopy
- •EBUS-TBNA
- •Ultrathin Bronchoscopy
- •Transbronchial Lung Cryobiobsy (TBLC)
- •Therapeutic Procedures Via FB
- •LASER Bronchoscopy
- •Electrocautery
- •Argon Plasma Coagulation (APC)
- •Cryotherapy
- •Photodynamic Therapy
- •Airway Stent Placement
- •Endobronchial Valve Placement
- •Conclusion
- •References
- •History and Historical Perspective
- •Indications and Contraindications
- •Procedure Description
- •Procedure Planning
- •Target Approximation
- •Sampling
- •Complications
- •Future Directions
- •Summary and Recommendations
- •References
- •4: Rigid Broncoscopy
- •Innovations
- •Ancillary Equipment
- •Rigid Bronchoscopy Applications
- •Laser Bronchoscopy
- •Tracheobronchial Prosthesis
- •Transbronchial Needle Aspiration (TBNA)
- •Rigid Bronchoscope in Other Treatments for Bronchial Obstruction
- •Mechanical Debridement
- •Pediatric Rigid Bronchoscopy
- •Tracheobronchial Dilatation
- •Foreign Bodies Removal
- •Other Indications
- •Complications
- •The Procedure
- •Some Conclusions
- •References
- •History and Historical Perspective
- •Indications and Contraindications
- •Preprocedural Evaluation and Preparation
- •Physical Examination
- •Procedure-Related Indications
- •Application of the Technique
- •Topical Anesthesia
- •Anesthesia of the Nasal Mucosa and Nasopharynx
- •Anesthesia of the Mouth and Oropharynx
- •Superior Laryngeal Nerve Block
- •Recurrent Laryngeal Nerve Block (RLN)
- •Conscious Sedation
- •Monitored Anesthesia Care (MAC)
- •General Anesthesia
- •Monitoring the Depth of Anesthesia
- •Interventional Bronchoscopy Suites
- •Airway Devices
- •Laryngeal Mask Airway (LMA)
- •Endotracheal Tube (ETT)
- •Rigid Bronchoscope
- •Modes of Ventilation
- •Spontaneous Ventilation
- •Assisted Ventilation
- •Noninvasive Positive Pressure Ventilation (NIV)
- •Positive Pressure Controlled Mechanical Ventilation
- •Jet Ventilation
- •Electronic Mechanical Jet Ventilation
- •Postprocedure Care
- •Special Consideration
- •Anesthesia for Peripheral Diagnostic and Therapeutic Bronchoscopy
- •Anesthesia for Interventional Bronchoscopic Procedures During the COVID-19 Pandemic
- •Summary and Recommendations
- •Conclusion
- •References
- •Background
- •Curricular Structure and Delivery
- •What Is a Bronchoscopy Curriculum?
- •Tradition, Teaching Styles, and Beliefs
- •Using Assessment Tools to Guide the Educational Process
- •The Ethics of Teaching
- •When Learners Teach: The Journey from Novice to Mastery and Back Again
- •The Future Is Now
- •References
- •Interventional Procedure
- •Assessment of Flow–Volume Curve
- •Dyspnea
- •Analysis of Pressure–Pressure Curve
- •Conclusions
- •References
- •Introduction
- •Adaptations of the IP Department
- •Environmental Control
- •Personal Protective Equipment
- •Procedure Performance
- •Bronchoscopy in Intubated Patients
- •Other Procedures in IP Unit
- •References
- •Introduction
- •Safety
- •Patient Safety
- •Provider Safety
- •Patient Selection and Screening
- •Lung Cancer Diagnosis and Staging
- •Inpatients
- •COVID-19 Clearance
- •COVID Clearance: A Role for Bronchoscopy
- •Long COVID: A Role for Bronchoscopy
- •Preparing for the Next Pandemic
- •References
- •Historical Perspective
- •Indications and Contraindications
- •Evidence-Based Review
- •Summary and Recommendations
- •References
- •Introduction
- •Clinical Presentation
- •Diagnosis
- •Treatment
- •History and Historical Perspectives
- •Indications and Contraindications
- •Benign and Malignant Tumors
- •Tumors with Uncertain Prognosis
- •Application of the Technique
- •Evidence Based Review
- •Summary and Recommendations
- •References
- •12: Cryotherapy and Cryospray
- •Introduction
- •Historical Perspective
- •Equipment
- •Cryoadhesion
- •Indications
- •Cryorecanalization
- •Cryoadhesion and Foreign Body Removal
- •Cryoadhesion and Mucus Plugs/Blood Clot Retrieval
- •Endobronchial Cryobiopsy
- •Transbronchial Cryobiopsy for Lung Cancer
- •Safety Concerns and Contraindications
- •Cryoablation
- •Indications
- •Evidence
- •Safety Concerns and Contraindications
- •Cryospray
- •Indications
- •Evidence
- •Safety Concerns and Contraindications
- •Advantages of Cryotherapy
- •Limitations
- •Future Research Directions
- •References
- •13: Brachytherapy
- •History and Historical Perspective
- •Indications and Contraindications
- •Application of the Technique
- •Evidence-Based Review
- •Adjuvant Treatment
- •Palliative Treatment
- •Complications
- •Summary and Recommendations
- •References
- •14: Photodynamic Therapy
- •Introduction
- •Photosensitizers
- •First-Generation Photosensitizers
- •M-Tetrahidroxofenil Cloro (mTHPC) (Foscan®)
- •PDT Reaction
- •Tumor Damage Process
- •Procedure
- •Indications
- •Curative PDT Indications
- •Palliative PDT Indications
- •Contraindications
- •Rationale for Use in Early-Stage Lung Cancer
- •Rationale
- •PDT in Combination with Other Techniques for Advanced-Stage Non-small Cell Lung Cancer
- •Commentary
- •Complementary Endoscopic Methods for PDT Applications
- •New Perspectives
- •Other PDT Applications
- •Conclusions
- •References
- •15: Benign Airways Stenosis
- •Etiology
- •Congenital Tracheal Stenosis
- •Iatrogenic
- •Infectious
- •Idiopathic Tracheal Stenosis
- •Distal Bronchial Stenosis
- •Diagnosis Methods
- •Patient History
- •Imaging Techniques
- •Bronchoscopy
- •Pulmonary Function Test
- •Treatment
- •Endoscopic Treatment
- •Dilatation
- •Laser Therapy
- •Stents
- •How to Proceed
- •Stent Placement
- •Placing a Montgomery T Tube
- •The Rule of Twos for Benign Tracheal Stenosis (Fig. 15.23)
- •Surgery
- •Summary and Recommendations
- •References
- •16: Endobronchial Prostheses
- •Introduction
- •Indications
- •Extrinsic Compression
- •Intraluminal Obstruction
- •Stump Fistulas
- •Esophago-respiratory Fistulas (ERF)
- •Expiratory Central Airway Collapse
- •Physiologic Rationale for Airway Stent Insertion
- •Stent Selection Criteria
- •Stent-Related Complications
- •Granulation Tissue
- •Stent Fracture
- •Migration
- •Contraindications
- •Follow-Up and Patient Education
- •References
- •Introduction
- •Overdiagnosis
- •False Positives
- •Radiation
- •Risk of Complications
- •Lung Cancer Screening Around the World
- •Incidental Lung Nodules
- •Management of Lung Nodules
- •References
- •Introduction
- •Minimally Invasive Procedures
- •Mediastinoscopy
- •CT-Guided Transthoracic Biopsy
- •Fluoroscopy-Guided Transthoracic Biopsies
- •US-Guided Transthoracic Biopsy
- •Thoracentesis and Pleural Biopsy
- •Thoracentesis
- •Pleural Biopsy
- •Surgical or Medical Thoracoscopy
- •Image-Guided Pleural Biopsy
- •Closed Pleural Biopsy
- •Image-Guided Biopsies for Extrathoracic Metastases
- •Tissue Acquisition, Handling and Processing
- •Implications of Tissue Acquisition
- •Guideline Recommendations for Tissue Acquisition in Mediastinal Staging
- •Methods to Overcome Challenges in Tissue Acquisition and Genotyping
- •Rapid on-Site Evaluation (ROSE)
- •Sensitive Genotyping Assays
- •Liquid Biopsy
- •Summary, Recommendations and Highlights
- •References
- •History
- •Data Source and Methodology
- •Tumor Size
- •Involvement of the Main Bronchus
- •Atelectasis/Pneumonitis
- •Nodal Staging
- •Proposal for the Revision of Stage Groupings
- •Small Cell Lung Cancer (SCLC)
- •Discussion
- •Methodology
- •T Descriptors
- •N Descriptors
- •M Descriptors
- •Summary
- •References
- •Introduction
- •Historical Perspective
- •Fluoroscopy
- •Radial EBUS Mini Probe (rEBUS)
- •Ultrasound Bronchoscope (EBUS)
- •Virtual Bronchoscopy
- •Trans-Parenchymal Access
- •Cone Beam CT (CBCT)
- •Lung Vision
- •Sampling Instruments
- •Conclusions
- •References
- •History and Historical Perspective
- •Narrow Band Imaging (NBI)
- •Dual Red Imaging (DRI)
- •Endobronchial Ultrasound (EBUS)
- •Optical Coherence Tomography (OCT)
- •Indications and Contraindications
- •Confocal Laser Endomicroscopy and Endocytoscopy
- •Raman Spectrophotometry
- •Application of the Technique
- •Supplemental Technology for Diagnostic Bronchoscopy
- •Evidence-Based Review
- •Summary and Recommendations, Highlight of the Developments During the Last Three Years (2013 on)
- •References
- •Introduction
- •History and Historical Perspective
- •Endoscopic AF-OCT System
- •Preclinical Studies
- •Clinical Studies
- •Lung Cancer
- •Asthma
- •Airway and Lumen Calibration
- •Obstructive Sleep Apnea
- •Future Applications
- •Summary
- •References
- •23: Endobronchial Ultrasound
- •History and Historical Perspective
- •Equipment
- •Technique
- •Indication, Application, and Evidence
- •Convex Probe Ultrasound
- •Equipment
- •Technique
- •Indication, Application, and Evidence
- •CP-EBUS for Malignant Mediastinal or Hilar Adenopathy
- •CP-EBUS for the Staging of Non-small Cell Lung Cancer
- •CP-EBUS for Restaging NSCLC After Neoadjuvant Chemotherapy
- •Complications
- •Summary
- •References
- •Introduction
- •What Is Electromagnetic Navigation?
- •SuperDimension Navigation System (EMN-SD)
- •Computerized Tomography
- •Computer Interphase
- •The Edge Catheter: Extended Working Channel (EWC)
- •Procedural Steps
- •Planning
- •Detecting Anatomical Landmarks
- •Pathway Planning
- •Saving the Plan and Exiting
- •Registration
- •Real-Time Navigation
- •SPiN System Veran Medical Technologies (EMN-VM)
- •Procedure
- •Planning
- •Navigation
- •Biopsy
- •Complications
- •Limitations
- •Summary
- •References
- •Introduction
- •Image Acquisition
- •Hardware
- •Practical Considerations
- •Radiation Dose
- •Mobile CT Studies
- •Future Directions
- •Conclusion
- •References
- •26: Robotic Assisted Bronchoscopy
- •Historical Perspective
- •Evidence-Based Review
- •Diagnostic Yield
- •Monarch RAB
- •Ion Endoluminal Robotic System
- •Summary
- •References
- •History and Historical Perspective
- •Indications and Contraindications
- •General
- •Application of the Technique
- •Preoperative Care
- •Patient’s Position and Operative Field
- •Incision and Initial Dissection
- •Palpation
- •Biopsy
- •Control of Haemostasis and Closure
- •Postoperative Care
- •Complications
- •Technical Variants
- •Extended Cervical Mediastinoscopy
- •Mediastinoscopic Biopsy of Scalene Lymph Nodes
- •Inferior Mediastinoscopy
- •Mediastino-Thoracoscopy
- •Video-Assisted Mediastinoscopic Lymphadenectomy
- •Transcervical Extended Mediastinal Lymphadenectomy
- •Evidence-Based Review
- •Summary and Recommendations
- •References
- •Introduction
- •Case 1
- •Adrenal and Hepatic Metastases
- •Brain
- •Bone
- •Case 1 Continued
- •Biomarkers
- •Case 1 Concluded
- •Case 2
- •Chest X-Ray
- •Computerized Tomography
- •Positive Emission Tomography
- •Magnetic Resonance Imaging
- •Endobronchial Ultrasound with Transbronchial Needle Aspiration
- •Transthoracic Needle Aspiration
- •Transbronchial Needle Aspiration
- •Endoscopic Ultrasound with Needle Aspiration
- •Combined EUS-FNA and EBUS-TBNA
- •Case 2 Concluded
- •Case 3
- •Standard Cervical Mediastinoscopy
- •Extended Cervical Mediastinoscopy
- •Anterior Mediastinoscopy
- •Video-Assisted Thoracic Surgery
- •Case 3 Concluded
- •Case 4
- •Summary
- •References
- •29: Pleural Anatomy
- •Pleural Embryonic Development
- •Pleural Histology
- •Cytological Characteristics
- •Mesothelial Cells Functions
- •Pleural Space Defense Mechanism
- •Pleura Macroscopic Anatomy
- •Visceral Pleura (Pleura Visceralis or Pulmonalis)
- •Parietal Pleura (Pleura Parietalis)
- •Costal Parietal Pleura (Costalis)
- •Pleural Cavity (Cavitas Thoracis)
- •Pleural Apex or Superior Pleural Sinus [12–15]
- •Anterior Costal-Phrenic Sinus or Cardio-Phrenic Sinus
- •Posterior Costal-Phrenic Sinus
- •Cost-Diaphragmatic Sinus or Lateral Cost-Phrenic Sinus
- •Fissures18
- •Pleural Vascularization
- •Parietal Pleura Lymphatic Drainage
- •Visceral Pleura Lymphatic Drainage
- •Pleural Innervation
- •References
- •30: Chest Ultrasound
- •Introduction
- •The Technique
- •The Normal Thorax
- •Chest Wall Pathology
- •Pleural Pathology
- •Pleural Thickening
- •Pneumothorax
- •Pulmonary Pathology
- •Extrathoracic Lymph Nodes
- •COVID and Chest Ultrasound
- •Conclusions
- •References
- •Introduction
- •History of Chest Tubes
- •Overview of Chest Tubes
- •Contraindications for Chest Tube Placement
- •Chest Tube Procedural Technique
- •Special Considerations
- •Pneumothorax
- •Empyema
- •Hemothorax
- •Chest Tube Size Considerations
- •Pleural Drainage Systems
- •History of and Introduction to Indwelling Pleural Catheters
- •Indications and Contraindications for IPC Placement
- •Special Considerations
- •Non-expandable Lung
- •Chylothorax
- •Pleurodesis
- •Follow-Up and IPC Removal
- •IPC-Related Complications and Management
- •Competency and Training
- •Summary
- •References
- •32: Empyema Thoracis
- •Historical Perspectives
- •Incidence
- •Epidemiology
- •Pathogenesis
- •Clinical Presentation
- •Radiologic Evaluation
- •Biochemical Analysis
- •Microbiology
- •Non-operative Management
- •Prognostication
- •Surgical Management
- •Survivorship
- •Summary and Recommendations
- •References
- •Evaluation
- •Initial Intervention
- •Pleural Interventions for Recurrent Symptomatic MPE
- •Especial Circumstances
- •References
- •34: Medical Thoracoscopy
- •Introduction
- •Diagnostic Indications for Medical Thoracoscopy
- •Lung Cancer
- •Mesothelioma
- •Other Tumors
- •Tuberculosis
- •Therapeutic Indications
- •Pleurodesis of Pneumothorax
- •Thoracoscopic Drainage
- •Drug Delivery
- •Procedural Safety and Contraindications
- •Equipment
- •Procedure
- •Pre-procedural Preparations and Considerations
- •Procedural Technique [32]
- •Medical Thoracoscopy Versus VATS
- •Conclusion
- •References
- •Historical Perspective
- •Indications and Contraindications
- •Evidence-Based Review
- •Endobronchial Valves
- •Airway Bypass Tracts
- •Coils
- •Other Methods of ELVR
- •Summary and Recommendations
- •References
- •36: Bronchial Thermoplasty
- •Introduction
- •Mechanism of Action
- •Trials
- •Long Term: Ten-Year Study
- •Patient Selection
- •Bronchial Thermoplasty Procedure
- •Equipment
- •Pre-procedure
- •Bronchoscopy
- •Post-procedure
- •Conclusion
- •References
- •Introduction
- •Bronchoalveolar Lavage (BAL)
- •Technical Aspects of BAL Procedure
- •ILD Cell Patterns and Diagnosis from BAL
- •Technical Advises for Conventional TLB and TLB-C in ILD
- •Future Directions
- •References
- •Introduction
- •The Pediatric Airway
- •Advanced Diagnostic Procedures
- •Endobronchial Ultrasound
- •Virtual Navigational Bronchoscopy
- •Cryobiopsy
- •Therapeutic Procedures
- •Dilation Procedures
- •Thermal Techniques
- •Mechanical Debridement
- •Endobronchial Airway Stents
- •Metallic Stents
- •Silastic Stents
- •Novel Stents
- •Endobronchial Valves
- •Bronchial Thermoplasty
- •Discussion
- •References
- •Introduction
- •Etiology
- •Congenital ADF
- •Malignant ADF
- •Cancer Treatment-Related ADF
- •Benign ADF
- •Iatrogenic ADF
- •Diagnosis
- •Treatment Options
- •Endoscopic Techniques
- •Stents
- •Clinical Results
- •Stent Complications
- •Other Available Stents
- •Other Endoscopic Methods
- •References
- •Introduction
- •Anatomy and Physiology of Swallowing
- •Functional Physiology of Swallowing
- •Epidemiology and Risk Factors
- •Types of Foreign Bodies
- •Organic
- •Inorganic
- •Mineral
- •Miscellaneous
- •Clinical Presentation
- •Acute FB
- •Retained FB
- •Radiologic Findings
- •Bronchoscopy
- •Airway Management
- •Rigid Vs. Flexible Bronchoscopy
- •Retrieval Procedure
- •Instruments
- •Grasping Forceps
- •Baskets
- •Balloons
- •Suction Instruments
- •Ablative Therapies
- •Cryotherapy
- •Laser Therapy
- •Electrocautery and APC
- •Surgical Management
- •Complications
- •Bleeding and Hemoptysis
- •Distal Airway Impaction
- •Iron Pill Aspiration
- •Follow-Up and Sequelae
- •Conclusion
- •References
- •Vascular Origin of Hemoptysis
- •History and Historical Perspective
- •Diagnostic Bronchoscopy
- •Therapeutic Bronchoscopy
- •General Measures
- •Therapeutic Bronchoscopy
- •Evidence-Based Review
- •Summary
- •Recommendations
- •References
- •History
- •“The Glottiscope” (1807)
- •“The Esophagoscope” (1895)
- •The Rigid Bronchoscope (1897–)
- •The Flexible Bronchoscope (1968–)
- •Transbronchial Lung Biopsy (1972) (Fig. 42.7)
- •Laser Therapy (1981–)
- •Endobronchial Stents (1990–)
- •Electromagnetic Navigation (2003–)
- •Bronchial Thermoplasty (2006–)
- •Endobronchial Microwave Therapy (2004–)
- •American Association for Bronchology and Interventional Pulmonology (AABIP) and Journal of Bronchology and Interventional Pulmonology (JOBIP) (1992–)
- •References
- •Index

172 |
E. Josan and J. Pannu |
|
|
are available in 1.9 and 2.4 mm size (Fig. 12.3a), for use with a minimum working channel of 2.0 and 2.8 mm, respectively [1]. The tip of the reusable cryoprobe is approximately 6 mm in length [7, 8]. The single use cryoprobes are exclusively used with ERBECRYO® 2 console and are developed to overcome technical limitations with miniaturization as well as to better ensure reproducibility. These disposable cryoprobes also overcome the risk of cross-contamination that can be an area of concern with reusable probes [9]. They are 115 cm in length and available in 1.1 mm, 1.7 mm and 2.4 mm (Fig. 12.3b), for use with a minimum working channel of 1.2, 2.0, and 2.8 mm, respectively [7].
Rigid cryoprobes are also available as straight or right-angled tip and may have a reheating system to allow rapid thawing. They are 60 cm in length with a 3 mm diameter and 9.2 mm cooling tip [1]. The equipment for spray cryotherapy is described in a separate section in the later part of this chapter.
Mechanism ofAction
The freezing effect of probe cryotherapy is based on the Joule-Thompson effect (Fig. 12.4). The pressurized cryogen gas from the cylinder (either carbon dioxide or nitrous oxide) is forced through the narrow inner channel of the exible cryoprobe to the tip of the probe. After passing the internal nozzle, the pressurized gas suddenly decompresses and cools. The relaxation of cryogen gathers energy from the surrounding area and cools down to the freezing temperature of the gas (−78.5 °C for CO2 and –89 °C for N2O) to incite freezing of tissue in contact with the tip of the probe [4]. Since it is a closed system, the cryogen does not come in direct contact with the tissue. The decompressed gas is then returned to the console via the external channel of cryoprobe which then dissipates into the surrounding atmosphere. In contrast, spray cryotherapy directly applies the liquid nitrogen to endobronchial tissue leading to a ash freeze. While the freezing temperature of CO2 is −78.5 °C [4], the value can be reduced via the cryotherapy system to achieve temperatures between −35 °C and −50 °C at the tip of the probe which is required for an effective cell death in neoplastic tissue [7, 10].
Fig. 12.4 Title: Joule-Thompson Effect. Description: Cryogen is released from the tank in a controlled fashion. It travels through the inner channel and is forced to pass through the nozzle at tip of the cryoprobe. The relaxation gather energy and cools down the surrounding region leading to a freezing effect. (Image© Erbe Elektromedizin GmbH)
Techniques andApplication
The tissue effects of cryotherapy can be broadly classifed into two categories based upon the underlying principle.
\1.\ Cryoadhesion uses the strong adherence of cryoprobe and target tissue.
\2.\ Cryoablation uses intracellular cell death from rapid freezing.
Cryoadhesion
Cryoadhesion works on the principle of freezing the uid between the tip of the cryoprobe and the target as well as the uid within the target. This leads to the formation of ice crystals and adheres
Данная книга находится в списке для перевода на русский язык сайта https://meduniver.com/
12 Cryotherapy and Cryospray |
173 |
|
|
a |
b |
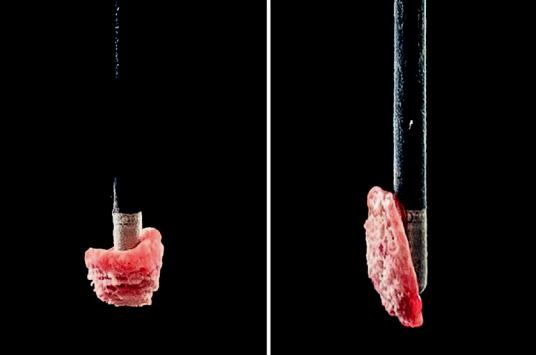
Fig. 12.5 Title: Cryoadhesion. Description: The target tissue can be adhered to the cryoprobe with either a “frontal” approach that leads to circumferential application (a)
or the “lateral” approach that can adhere a longer segment (b). (Image© Erbe Elektromedizin GmbH)
the two surfaces together. This effect can be used with either a frontal or tangential approach (Fig. 12.5) to remove blood clots, extract foreign bodies, obtain endobronchial or transbronchial tissue biopsies, and recanalize the airway by extracting endobronchial tumors [7].
This cryoadhesive effect is dependent on the size of the frozen area which is in turn affected by the freeze time, contact area, moisture level, tissue/foreign body properties, the cylinder pressure, and ambient temperature of the environment [7]. Freeze time is the easiest to control by the operator. There is rapid freezing in the frst 5 seconds following which the effect is gradually weaned due to freezing around the probe tip and thermal equilibrium which dissipates the surrounding heat to the frozen probe tip [7]. The freeze time can be controlled by allocating a preset or by pressing the pedal for longer period in the “free freeze” preset. The contact area is determined by the diameter of the probe tip with larger freezing effect exerted by the larger probe. The
1.7, 1.9, and 2.4 mm probes are adequate for both endobronchial and transbronchial use.
Moisture between the tip and target as well within the target plays a vital role for cryoadhesion. An organic material with good water content such as lung tissue, tumor, or a porous foreign body is more likely to freeze on contact with cryoprobe in comparison to an inorganic object such as metallic or plastic foreign body. Different body tissues respond differently to cryotherapy with some tissues being sensitive due to their water content (e.g., tumor, granulation tissue, mucus membrane). On the other hand, some tissues are resistant to effect of cryotherapy (e.g., cartilage, fat, connective tissue and fat). Therefore, the tracheobronchial wall which is mainly composed of fbrocartilaginous structure is less likely to be damaged from the effects of repeated application of cryotherapy, while a tumor attached to this wall will be affected signifcantly [1]. The ambient temperature of the working environment has a direct effect on the
174 |
E. Josan and J. Pannu |
|
|
pressure in the cryogen cylinder which can affect the performance of the machine. High environmental temperature can lead to high cylinder pressure which affects the evaporation pressure of liquid CO2. While a high pressure can be compensated by the machine, the low pressure can lead to poor freezing. Newer devices can alert the user of such issues [7].
Indications
The exible cryotherapy probes are widely used upon the principle of cryoadhesion. The effect is useful for recanalization of central airway obstruction (whether malignant or benign); a tangential biopsy of infltrating tumors, devitalizing tissue; removal of blood clots or foreign body; and for transbronchial biopsy in interstitial lung disease or peripheral lung nodules [9, 11]. The use of cryotherapy for these purposes of tumor debulking, endobronchial cryobiopsy, and cryorecanalization is endorsed by British thoracic society [12] and American College of Chest Physicians [13].
Cryotherapy is often used in conjunction with thermal therapies such as electrocautery, laser, argon plasma coagulation, balloon dilation, and airway stenting to restore and maintain airway patency. Unlike thermal therapy, it is safe to use with higher oxygen concentration and the preferred therapy when low fraction of inspired oxygen (FiO2) cannot be tolerated by the patient. It is also safer to use around combustible substances such as stents and endotracheal tubes. Moreover, it does not interfere with cardiac pacemakers or implanted defbrillators unlike electrical therapy [1].
Cryorecanalization
Patient with endobronchial tumor and airway obstruction can beneft from endoscopic debulking if the airway and parenchyma distal to this obstruction are salvageable. The principle of cryoadhesion can be used to remove exophytic endoluminal tumor or granulation tissue for an immediate effect on airway patency. The cryoprobe is activated on contact with target tissue for 3–15 seconds to incite cryoadhesion followed by a rapid pull with the intent of removing large
pieces of tissue (Fig. 12.6). Since the debulked tissue is too large for working channel of bronchoscope, the probe is removed en-bloc and thawed in saline [14]. The bronchoscope should be quickly reinserted to assess any bleeding from the site. For a central tumor, any size cryoprobe can be used depending on the intended size of tissue fragments. Typically, the larger probes (1.7, 1.9, and 2.4 mm) are utilized for central cryodebulking due to larger size effects which lead to more effcient tumor removal and recanalization and yield larger tissue fragments for pathological testing [7]. This method is safe and effective for rapid debulking of endobronchial tumor and restores airway patency more rapidly than its counterpart cryodevitalization.
The goal of cryorecanalization (also referred to as cryodebulking) is to improve the patient’s performance status and survival even if they are not eligible for surgical treatment [12]. The overall effcacy of cryorecanalization in symptom palliation is reported in 70–90% patients [14]. The largest analysis on cryorecanalization by Maiwand et al. (n = 476) reported a mean of 2.4 cryosurgical treatments in malignant endobronchial tumors. The study reported that 86% had improvement in ≥1 symptoms (hemoptysis, cough, dyspnea, and chest pain). The mean Karnofsky score improved from 59.6 to 75.2 and the average increase in Forced expiratory volume in the frst second (FEV1) and Forced Vital Capacity (FVC) was 90 and 130 mL, respectively [14, 15].
In addition, Schumann et al. (n = 225) described the use of cryorecanalization in symptomatic airway stenosis and noted a 91.1% success rate. In this retrospective analysis, length of lesion more than 2 cm was associated with unsuccessful intervention. Adjunctive modalities such as stent (4.9%) or APC (16.4%) were used infrequently [16]. Another retrospective analysis by Inaty et al. (n = 156) reported restoration of airway patency in 95% patients with improvement in respiratory symptoms noted in 82% of symptomatic patients. Adjunctive modalities such as mechanical debridement (51%) and thermal therapy (EC 30%, APC 17%) were used much more frequently in this study. They noted cryotherapy
Данная книга находится в списке для перевода на русский язык сайта https://meduniver.com/

12 Cryotherapy and Cryospray |
175 |
|
|
a |
b |
c |
d |
Fig. 12.6 Title: Cryorecanalization. Description: (a) shows a necrotic tumor in left mainstem orifce resulting in complete obstruction. (b) Shows the effect of partial removal of this tumor using cryodebulking technique. (c) Shows the result of complete cryorecanalization with res-
toration of patency of left mainstem bronchus. (d) Shows a follow up bronchoscopy at 4 weeks. (Images courtesy of Dr. Nicholas Pastis, The Ohio State University Hospital, Columbus, Ohio)
was most effcacious in treating central airway lesion [17]. A smaller prospective study by Hetzel et al. (n = 60) reported successful recanalization in 83% out of which 61% had complete and 22% had partial improvement in patency [5]. Another small study byYilmaz et al. (n = 40) reported successful cryorecanalization in 72.5% patients. They also reported that the success rate was related to the presence of the distal involvement and the older age of obstruction [18].
Cryoadhesion and Foreign Body Removal
Foreign body aspiration is common in children younger than 3 years old and in adults after sixth decade of life. Flexible bronchoscopy has gained signifcant experience with various apparatuses available for endobronchial use. It has therefore replaced rigid bronchoscopy as a less invasive alternative [19]. When exible bronchoscopy
