
Фізична та колоїдна хімія
.pdf/ 10. ; } /</-; (; G/
#$, % $%, " - , + / ( , + /-( . $ ($ "% + /" )&, -+ $ . ! + % - % ( " ) - ) ) , " $ - , + / . * , / ) , ( -+ i . + ’ ( + / + // ) , / / ( ( * .
) $ ) -$, $% - .
10.1. 9%#& " ?$ ! * # . ( ) ) ! !$"- ' ! * #
D ; + , -) , $ "
& (%'D), ; + /" -*$ . $ -) ) ) "( . « % $»). ) -) . * , -, $ , + " $ , , -) , + $ .
J % ) ( - /" , $ % - ($ #I . 37. (:
191
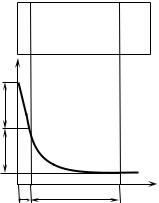
) – ) $. 0 - " $. * , / ) , + /" -$ ) ) $ " "$ + ) $ $ .
& ) + , ) ( +: ( ) ( " -$ , " $ -. C- ) "+ ) $ $ + $$ . , " ( -$ / -$ .
& ) . +$$% , ( $ - " ’ $ + -/ . B -) /" , ( +-$ ( ( " +- . ) + $ ) - , / $ $ ($.
? + ( $/ - / . 0 - ) ( , " -, ; /" /. -+ / ) ) /– , -/ $$ , / /" . ) ) -
" :H0 / H2 ,Pt , ) ( " /
+- $ $.
$( ) , , / ) ,; $ )" ) :
193
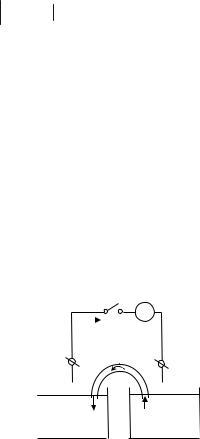
( )Pt, H2 H 
 Cu2 Cu( ) .
Cu2 Cu( ) .
# / # ? ) . J ) ) " , / /, ;
/" |
1 ( H = 1 /), " |
||
PH2 = 1 |
K. |
|
|
C ) |
" , |
|
|
/" , |
– |
||
, ) // $ $. |
|||
10.2. F# # , ! ! ! +# ! )- $? |
|
||
G- |
) ($ |
) – |
|
) , $ $ $ $+ / $% ) % . ?$ + ( -
-D + |
|
|
|
|
A |
|||
. 38. * - |
|
|
c |
|
|
|
|
|
" % % |
|
|
|
K+Cl– |
|
|
|
|
, - |
– |
+ |
||||||
$ |
|
|
|
|
|
|
|
|
. ’" ( |
|
Zn |
|
|
|
Cu |
|
|
+ / KCl, |
|
|
|
|
|
|||
|
|
|
|
|
|
|
||
|
+ " - |
Zn2+ + SO42– |
u2+ + SO42– |
|||||
|
+ ) " |
|
|
|
|
|
|
|
% ( . |
; . 38. F# # , ! ! ! |
D ; |
+# ! )–$? |
/), " - $ – , +
) " / . 0 " ( ) .
J ) $ $ + -$ $ $ " + .
194

] - ? |
|
|||
- , : |
- |
|||
– , |
– . |
|
||
W ) ) –D + " - |
||||
. C , + ) ( ), |
- |
|||
$/: |
Zn2 2e . |
|
|
|
Zn0 |
|
|
||
C , + ( ) |
- |
|||
$/: |
|
|
|
|
Cu 2 2e |
Cu 0 . |
|
|
|
? , $ ) - |
||||
, " ) : |
|
|
|
|
Zn0 Cu 2 |
Zn2 Cu0 . |
|
|
|
, / + / + ) )" -
. |
|
|
|
?$ ) |
–D + ( - |
||
+ : |
|
|
|
(–)Zn | Zn2+ || Cu2+ |Cu(+) |
|||
|
|
|
|
¯Zn2+/Zn |
¯ |
|
¯Cu2+/Cu |
|
|||
C $ $ -
:
| – ( « – »;
||– ( ZnSO4 CuSO4;
:Zn2 / Zn ,:Cu2 / Cu – + )
;
:K – ( $ - , " $ % $ .
& / ) ( )-/ , ( - , KCl + KNO3).
195
0 # ? ) ) –D + ($ /:
E :Cu2 / Cu :Zn2 / Zn : "“z ;: "“z 9 0 .
' –D + $ )$ ’" ( - ), " $ % . E- / ( + ) ’" ( + - ); $ $ + -.
J +- # ? ) ) - / / :
E:K :A .
10.3./ !$" " K , # # '# # ' ! ! ! #. ;- ) ) !" #
* ( , / - ( . ) ) ,$ $ $ $. 0 " ' ++ –' ) ( . 3) Wƒmax ) Wƒmax = n·F·E;' ++ –' ) ( :
|
|
|
|
E |
(10.2) |
n F E H T n F |
. |
|
|
T P |
|
J © $% (10.2) #-#$ $ $ . ! n – , ; + /-i i %.
0 ($ |
|
|
E |
/ - |
|
0 |
|
|
|
||
|
|||||
|
|
|
|
||
|
|
|
T P |
|
|
')( $ $ . & ( 0+ ) + ) " ) / (0 > 0), + " %% (0 < 0), + /" $ )$% % (0 = 0).
, ;
196
|
|
/ |
|
WMAX |
G H T S H T |
WMAX |
, |
/ |
|
|
|
|
|
T P |
|
, $/ (7.2), ( - :
|
|
|
|
|
|
|
|
|
|
|
|
' S |
|
E |
|
|
|
|
|
||||
n F |
|
|
|
|
|
|
|
||||
' |
|
|
|
T P |
|
|
|
||||
' |
|
|
|
|
|
|
|
|
E |
|
|
' |
|
|
|
|
|
|
|
|
|||
H |
n F E |
|
|
|
|
||||||
|
|
|
|||||||||
T n F |
|
|
(10.3) |
||||||||
' |
|
|
|
|
|
|
|
|
T P |
||
' |
|
|
G |
0 |
|
n F E |
|
|
|
|
|
' ln K |
a |
|
|
|
|
|
|
||||
|
|
|
|
|
|||||||
' |
|
RT |
|
|
|
RT |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
) ) $% - % 0-' ( )
G WMAX/ RT ln Ka RT ln @ a n F E , (10.4)"
E |
RT |
ln Ka |
|
RT |
ln @ a . |
(10.5) |
|
|
|||||
|
nF |
|
nF |
|
||
, ; $ , +-$ %, // 1. ! ln a = 0. 0:
|
RT |
ln Ka E 0 . |
(10.6) |
||
|
nF |
||||
|
|
|
|
|
|
! (10.5) ( : |
|
||||
E E0 |
RT |
ln@a , |
(10.7) |
||
|
|||||
|
|
|
nF |
|
|
|
|
|
|
|
|
E0 :K0 :A0 . |
|
||||
) ) –D + , |
|||||
$ $ : |
|
|
|||
Zn0 Cu2 |
|
Zn2 Cu0 . |
|
||
, ; + /- i i %, n = 2, " , %$ ". ! (10.7) " :
197
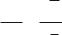

2. % ! Cd2+ | Cd(Hg).
# " ) : Cd2+ + 2e = Cd0.) $ /:
0 |
|
|
|
RT |
aCd 2 |
|
||||
:Cd 2 |
|
Ccd (Hg ) :Cd 2 |
|
Cd (Hg ) |
|
|
ln |
|
. |
(10.16) |
|
|
|
|
|||||||
|
|
|
|
|
|
2F |
aCd (Hg ) |
|
||
, / + / " , ; ) " / Cd2+ , - / Cd ) (aCd(Hg)).
3. e
E ) ) , + + . 0 / - ) , + " ) + , ; ) ) . ! "
|
$ |
. |
: |
|
(H+ | H, Pt) $ |
H+ + 1e = ½ H2, ) " : |
||
: 0 |
|
H2 |
,Pt |
0;ln a 2,3lg a; pH lg[a |
]; p |
1 . |
H |
|
H |
|
H2 |
:H |
|
H2 ,Pt |
2,3lg aH 2,3lg pH . |
(10.17) |
||||||
|
||||||||||
: |
|
(Cl– | |
Cl2, Pt) $ |
|
||||||
½ Cl2 + 1e = Cl–, ) : |
|
|||||||||
|
|
:Cl |
|
:Cl0 |
|
|
RT |
ln aCl . |
(10.18) |
|
|
|
Cl2 ,Pt |
Cl2 ,Pt |
|
||||||
|
|
|
|
|
|
|
|
F |
|
|
$) " / - . C , +$ , -$) " ; - % //$ .
' $ $ . ) -, / , ) (- ) ( – ) - ), , ( % .
200
