|
Indium
Indium is a chemical element with symbol In and atomic number 49. This rare, very soft, malleable and easily fusible poor metal is chemically similar to gallium and thallium, and shows intermediate properties between these two. Indium was discovered in 1863 and named for the indigo blue line in its spectrum that was the first indication of its existence in zinc ores, as a new and unknown element. The metal was first isolated in the following year. Zinc ores continue to be the primary source of indium, where it is found in compound form. Very rarely the element can be found as grains of native (free) metal, but these are not of commercial importance. Indium's current primary application is to form transparent electrodes from indium tin oxide (ITO) in liquid crystal displays and touchscreens, and this use largely determines its global mining production. It is widely used in thin-films to form lubricated layers (during World War II it was widely used to coat bearings in high-performance aircraft). It is also used for making particularly low melting point alloys, and is a component in some lead-free solders. Indium is not known to be used by any organism. In a similar way to aluminium salts, indium(III) ions can be toxic to the kidney when given by injection, but oral indium compounds do not have the chronic toxicity of salts of heavy metals, probably due to poor absorption in basic conditions. Radioactive indium-111 (in very small amounts on a chemical basis) is used in nuclear medicine tests, as a radiotracer to follow the movement of labeled proteins and white blood cells in the body. Characteristics Physical
Indium wetting the glass surface of a test tube Indium is a very soft, silvery-white, relatively rare poor metal with a bright luster. Like tin, when it is bent indium emits a high-pitched "cry".[3] Like gallium, indium is able to wet glass. Like both, indium has a low melting point, 156.60 °C (313.88 °F); higher than its lighter homologue, gallium, but lower than its heavier homologue, thallium, and lower than tin.[4] Its boiling point is, however, moderate, being 2072 °C (3762 °F), which is higher than that of thallium, but lower than that of gallium, showing opposition to melting points trend. The density of indium, 7.31 g·cm−3, is also higher than that of gallium, but lower than that of thallium.[4] An indium atom has 49 electrons, having an electronic configuration of [Kr]4d105s25p1. In its compounds, indium most commonly loses its three outermost electrons, becoming indium(III) ions, In3+, but in some cases the pair of 5s-electrons can stay within the atom, indium thus oxidized only to indium(I), In+. This happens due to inert pair effect, which occurs because of stabilization of 5s-orbital due to relativistic effects, which are stronger closer to the bottom of the periodic table. Thallium shows an even stronger effect, making oxidation to thallium(I) more likely than to thallium(III), making +1 the more likely oxidation state.[5] A number of standard electrode potentials, depending on the reaction under study,[6] are reported for indium:
Chemical Indium is a poor metal and chemically, is the intermediate element between its group 13 neighbors gallium and thallium. It shows two main oxidation states, which are +1 and +3, with latter being more stable, whereas the only common oxidation state of gallium is +3 and thallium shows +1 more likely than +3, with thallium(III) being a moderately strong oxidizing agent, while indium(III) is stable and indium(I) is a powerful reducing agent.[7] Indium does not react with water, but it is oxidized by stronger oxidizing agents, such as halogens or oxalic acid, to give indium(III) compounds. It does not react with boron, silicon or carbon, and the corresponding boride, silicide or carbide are not known. Similarly, reaction between indium and hydrogen has not been observed, but both indium(I) and indium(III) hydrides are known.[8] Indium(III) oxide is formed at high temperatures during reaction between indium and oxygen, with blue flame. It is amphoteric, i. e. it can react with both acids and bases. Its reaction with water results in insoluble indium(III) hydroxide, which is also amphoteric, reacting with alkalies to give indates(III) and with acids to give indium(III) salts: In(OH)3 + 2 NaOH → 2 Na[InO2] + H2O In(OH)3 + 3 HCl → InCl3 + 3 H2O The hydrolysis of sodium indate(III) gives weak indic acid, HInO2. Out of common indium(III) salts, chloride, sulfate and nitrate are soluble. In water solutions, In3+ and [InO2]- ions are hydrolyzed to give InOH2+ and HInO2 due to generally amphoteric character of indium(III) ions. Indium(III) compounds are not well-soluble, similarly to thallium(III) compounds; however, indium(III) salts of strong acids, such as chloride, sulfate and nitrate are soluble, hydrolyzing in water solutions. The In3+ ion is colorless in solution because of the absence of unpaired electrons in the d- and f-electron shells.[7] Indium(I) compounds are not as common as indium(III) ones; only chloride, bromide, iodide, sulfide and cyclopentadienyl are well-characterized. Indium(I) sulfide is the product of reaction between indium and sulfur or indium and hydrogen sulfide, and can be received at 700—1000 °C. Indium(I) oxide black powder is received at 850 °C during reaction between indium and carbon dioxide or during decomposition of indium(III) oxide at 1200 °C. Cyclopentadienylindium(I), which was the first organoindium(I) compound reported,[9] is polymer consisting of zigzag chains of alternating indium atoms and cyclopentadienyl complexes. Less frequently, indium shows intermediate oxidation state +2, which lies between the common ones, most notably in halides, In2X4 and [In2X6]2-.[10] Several other compounds are known to combine indium(I) and indium(III), such as InI6(InIIICl6)Cl3,[11] InI5(InIIIBr4)2(InIIIBr6),[12] InIInIIIBr4.[10] Isotopes Main article: Isotopes of indium Indium occurs naturally on Earth only in two primordial nuclides, indium-113 and indium-115. Out of this two, indium-115 makes up 95.7% of all indium but it is radioactive, decaying to tin-115 via beta decay with half-life of 4.41×1014 years, four orders of magnitude larger than the age of the universe and nearly 50,000 times longer than that of natural thorium.[13] This situation is uncommon among stable chemical elements; only indium, tellurium, and rhenium have been shown to have most-abundant isotopes that are radioactive. The less common natural isotope of indium, indium-113, is stable. Indium has 39 known isotopes, ranging in mass between 97 and 135. Only one of them is stable and one has half-life exceeding 1014 years; the most stable other indium isotope is indium-111, which has half-life of approximately 2.8 days. All other isotopes have half-lives shorter than 5 hours. Indium also has 47 meta states, out of which indium-114m1 is the most stable, being more stable than ground state of any indium isotope, except for the primordial ones. Creation and occurrence See also category: Indium compounds
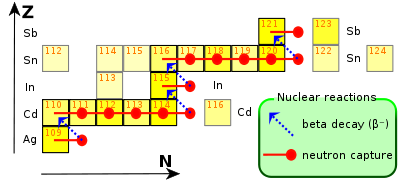
The s-process acting in the range from silver to antimony. Indium is created via the long-lasting, (up to thousands of years), s-process in low-medium mass stars (which range in mass between 0.6 and 10 masses of Sun). When a silver-109 atom, which comprises approximately half of all silver in existence, catches a neutron, it undergoes a beta decay to become cadmium-110. Capturing further neutrons, it becomes cadmium-115, which decays to indium-115 via another beta decay. This explains why the radioactive isotope predominates in abundance compared to the stable one.[14] Indium is 61st most abundant element in the Earth's crust at approximately 49 ppb, making indium approximately as abundant as mercury.[15] Fewer than 10 indium minerals are known, such as dzhalindite (In(OH)3) and indite (FeIn2S4),[16] but none of these occurs in significant deposits. Based on content of indium in zinc ore stocks, there is a worldwide reserve base of approximately 6,000 tonnes of economically viable indium.[17] However, the Indium Corporation, the largest processor of indium, claims that, on the basis of increasing recovery yields during extraction, recovery from a wider range of base metals (including tin, copper and other polymetallic deposits) and new mining investments, the long-term supply of indium is sustainable, reliable and sufficient to meet increasing future demands.[18] This conclusion may be reasonable considering that silver, which is one-third as abundant as indium in the Earth's crust,[19] is currently mined at approximately 18,300 tonnes per year,[20] which is 40 times greater than current indium mining rates. History In 1863, the German chemists Ferdinand Reich and Hieronymous Theodor Richter were testing ores from the mines around Freiberg, Saxony. They dissolved the minerals pyrite, arsenopyrite, galena and sphalerite in hydrochloric acid and distilled raw zinc chloride. As it was known that ores from that region sometimes contain thallium they searched for the green emission lines with spectroscopy. The green lines were absent but a blue line was present in the spectrum. As no element was known with a bright blue emission they concluded that a new element was present in the minerals. They named the element with the blue spectral line indium, from the indigo color seen in its spectrum.[21][22] Richter went on to isolate the metal in 1864.[23] At the World Fair 1867 an ingot of 0.5 kg (1.1 lb) was presented.[24] In 1924, indium was found to have a valuable ability to stabilize non-ferrous metals, which was the first significant use for the element.[25] It took until 1936 for the U.S. Bureau of Mines to list indium as a commodity, and even in early the 1950s only very limited applications for indium were known, the most important of which was making light-emitting diodes and coating bearings for aircraft engines during World War II. The start of production of indium-containing semiconductors started in 1952. The development and widespread use of indium-containing nuclear control rods increased demand during the 1970s, and the use of indium tin oxide in liquid crystal displays increased and became the major application by 1992.[26][27][28][29][30] Production
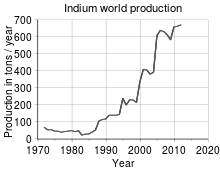
World production trend [31] The lack of indium mineral deposits and the fact that indium is enriched in sulfidic lead, tin, copper, iron and predominately in zinc deposits, makes zinc production the main source for indium. The indium is leached from slag and dust of zinc production. Further purification is done by electrolysis. The exact process varies with the exact composition of the slag and dust.[3][24] Indium is produced mainly from residues generated during zinc ore processing but is also found in iron, lead, and copper ores.[3] China is a leading producer of indium (390 tonnes in 2012), followed by Canada, Japan and South Korea with 70 tonnes each.[32] The Teck Cominco refinery in Trail, British Columbia, is a large single-source indium producer, with an output of 32.5 tonnes in 2005, 41.8 tonnes in 2004 and 36.1 tonnes in 2003. South American Silver Corporation's Malku Khota property in Bolivia is a large resource of indium with an indicated resource of 1,481 tonnes and inferred resource of 935 tonnes.[33] Adex Mining Inc.’s Mount Pleasant Mine in New Brunswick, Canada, holds some of the world’s total known indium resources.[34] The amount of indium consumed is largely a function of worldwide LCD production. Worldwide production is currently 475 tonnes per year from mining and a further 650 tonnes per year from recycling.[18] Demand has risen rapidly in recent years with the popularity of LCD computer monitors and television sets, which now account for 50% of indium consumption.[35] Increased manufacturing efficiency and recycling (especially in Japan) maintain a balance between demand and supply. According to the UNEP, indium's end-of-life recycling rate is less than 1%.[36] Demand increased as the metal is used in LCDs and televisions, and supply decreased when a number of Chinese mining concerns stopped extracting indium from their zinc tailings. In 2002, the price was US$94 per kilogram. The recent changes in demand and supply have resulted in high and fluctuating prices of indium, which from 2006 to 2009 ranged from US$382/kg to US$918/kg. It has been estimated that there is less than 20 years left of indium supplies, based on current rates of extraction, demonstrating the need for additional recycling.[37] Applications
A magnified image of an LCD screen showing RGB pixels. Individual transistors are seen as white dots in the bottom part. The first large-scale application for indium was as a coating for bearings in high-performance aircraft engines during World War II. Afterward, production gradually increased as new uses were found in fusible alloys, solders, and electronics. In the 1950s, tiny beads of it were used for the emitters and collectors of PNP alloy junction transistors. In the middle and late 1980s, the development of indium phosphide semiconductors and indium tin oxide thin films for liquid crystal displays (LCD) aroused much interest. By 1992, the thin-film application had become the largest end use.[38][39] Electronics
Metal and alloys
Ductile indium wire
Other uses
Precautions and health issues Pure indium in metal form is considered nontoxic by most sources. In the welding and semiconductor industries, where indium exposure is relatively high, there have been no reports of any toxic side-effects. Indium compounds, like aluminum compounds, complex with hydroxyls to form insoluble salts in basic conditions, and are thus not well-absorbed from food, giving them fairly low oral toxicty. Soluble indium(III) is toxic when delivered parenterally, however, causing damage primarily to the kidney (both inner and outer parts), but additionally to heart and liver, and may be teratogenic.[56] Other indium compounds are toxic when administered outside the gastrointestinal tract: for example, anhydrous indium trichloride (InCl3) and indium phosphide (InP) are quite toxic when delivered into the lungs (the latter is a suspected carcinogen).[57][58] Occupational exposure to indium compounds was associated with PAP, cholesterol ester crystals and granulomas, pulmonary fibrosis, emphysema, and pneumothoraces. The available evidence suggests exposure to indium compounds causes a novel lung disease that may begin with PAP and progress to include fibrosis and emphysema, and, in some cases, premature death [59]
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Thallium
|
Thallium |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
81Tl |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Thallium in the periodic table |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Appearance |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
silvery
white
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
General properties |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Name, symbol, number |
thallium, Tl, 81 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Pronunciation |
/ˈθæliəm/ THAL-ee-əm |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Element category |
poor metal |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Group, period, block |
13, 6, p |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Standard atomic weight |
204.38(1) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Electron configuration |
[Xe] 4f14 5d10 6s2 6p1 2, 8, 18, 32, 18, 3
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
History |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Discovery |
William Crookes (1861) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
First isolation |
Claude-Auguste Lamy (1862) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Physical properties |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Phase |
solid |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Density (near r.t.) |
11.85 g·cm−3 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Liquid density at m.p. |
11.22 g·cm−3 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Melting point |
577 K, 304 °C, 579 °F |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Boiling point |
1746 K, 1473 °C, 2683 °F |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Heat of fusion |
4.14 kJ·mol−1 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Heat of vaporization |
165 kJ·mol−1 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Molar heat capacity |
26.32 J·mol−1·K−1 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Vapor pressure |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Atomic properties |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Oxidation states |
3, 2, 1 (mildly basic oxide) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Electronegativity |
1.62 (Pauling scale) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Ionization energies |
1st: 589.4 kJ·mol−1 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
2nd: 1971 kJ·mol−1 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
3rd: 2878 kJ·mol−1 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Atomic radius |
170 pm |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Covalent radius |
145±7 pm |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Van der Waals radius |
196 pm |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Miscellanea |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Crystal structure |
hexagonal close-packed
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Magnetic ordering |
diamagnetic[1] |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Electrical resistivity |
(20 °C) 0.18 µΩ·m |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Thermal conductivity |
46.1 W·m−1·K−1 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Thermal expansion |
(25 °C) 29.9 µm·m−1·K−1 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Speed of sound (thin rod) |
(20 °C) 818 m·s−1 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Young's modulus |
8 GPa |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Shear modulus |
2.8 GPa |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Bulk modulus |
43 GPa |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Poisson ratio |
0.45 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Mohs hardness |
1.2 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Brinell hardness |
26.4 MPa |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
CAS registry number |
7440-28-0 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Most stable isotopes |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Main article: Isotopes of thallium |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Thallium is a chemical element with symbol Tl and atomic number 81. This soft gray poor metal is not found free in nature. When isolated, it resembles tin, but discolors when exposed to air. Chemists William Crookes and Claude-Auguste Lamy discovered thallium independently in 1861, in residues of sulfuric acid production. Both used the newly developed method of flame spectroscopy, in which thallium produces a notable green spectral line. Thallium, from Greek θαλλός, thallos, meaning "a green shoot or twig," was named by Crookes. It was isolated by electrolysis a year later, by Lamy.
Thallium tends to oxidize to the +3 and +1 oxidation states as ionic salts. The +3 state resembles that of the other elements in thallium's group (boron, aluminum, gallium, indium). However, the +1 state, which is far more prominent in thallium than the elements above it, recalls the chemistry of alkali metals, and thallium(I) ions are found geologically mostly in potassium-based ores, and (when ingested) are handled in many ways like potassium ions (K+) by ion pumps in living cells.
Commercially, however, thallium is produced not from potassium ores, but as a byproduct from refining of heavy metal sulfide ores. Approximately 60–70% of thallium production is used in the electronics industry, and the remainder is used in the pharmaceutical industry and in glass manufacturing.[2] It is also used in infrared detectors. The radioisotope thallium-201 (as the soluble chloride TlCl) is used in small, nontoxic amounts as an agent in a nuclear medicine scan, during one type of nuclear cardiac stress test.
Soluble thallium salts (many of which are nearly tasteless) are highly toxic in quantity, and were historically used in rat poisons and insecticides. Use of these compounds has been restricted or banned in many countries, because of their nonselective toxicity. Notably, thallium poisoning results in hair loss. Because of its historic popularity as a murder weapon, thallium has gained notoriety as "the poisoner's poison" and "inheritance powder" (alongside arsenic).[3]
Contents
-
1 Characteristics
-
1.1 Isotopes
-
1.2 Chemistry
-
-
2 History
-
3 Occurrence and production
-
4 Applications
-
4.1 Historic uses
-
4.2 Optics
-
4.3 Electronics
-
4.4 High-temperature superconductivity
-
4.5 Medical
-
4.5.1 Thallium stress test
-
-
4.6 Other uses
-
-
5 Toxicity
-
6 Treatment and internal decontamination
-
7 Thallium pollution
-
8 See also
-
9 References
-
10 External links
Characteristics
Thallium is extremely soft, malleable and sectile enough to be cut with a knife at room temperature. It has a metallic luster that, when exposed to air, quickly tarnishes to a bluish-gray tinge, resembling lead. It may be preserved by immersion in oil. A heavy layer of oxide builds up on thallium if left in air. In the presence of water, thallium hydroxide is formed. Sulfuric and nitric acid dissolve thallium rapidly to make the sulfate and nitrate salts, while hydrochloric acid forms an insoluble thallium(I) chloride layer.[4] Its standard electrode potential is −0.34, slightly higher than the potential for iron (at −0.44).
Isotopes
Main article: Isotopes of thallium
Thallium has 25 isotopes which have atomic masses that range from 184 to 210. 203Tl and 205Tl are the only stable isotopes, and 204Tl is the most stable radioisotope, with a half-life of 3.78 years.[5]
202Tl (half-life 12.23 days) can be made in a cyclotron,[6] while 204Tl is made by the neutron activation of stable thallium in a nuclear reactor.[5][5][7]
201Tl (half-life 73 hrs), decays by electron capture, emitting Hg X-rays (~70–80 keV), and photons of 135 and 167 keV in 10% total abundance;[5] therefore it has good imaging characteristics without excessive patient radiation dose. It is the most popular isotope used for thallium nuclear cardiac stress tests.[8]
208Tl (half-life 3.05 minutes) is generated in the naturally-occurring thorium decay chain. Its prominent 2615 keV gamma ray is the dominant high-energy feature observed in natural background radiation.