
- •Contents
- •Contributors
- •Part I General Principles of Cell Death
- •1 Human Caspases – Apoptosis and Inflammation Signaling Proteases
- •1.1. Apoptosis and limited proteolysis
- •1.2. Caspase evolution
- •2. ACTIVATION MECHANISMS
- •2.2. The activation platforms
- •2.4. Proteolytic maturation
- •3. CASPASE SUBSTRATES
- •4. REGULATION BY NATURAL INHIBITORS
- •REFERENCES
- •2 Inhibitor of Apoptosis Proteins
- •2. CELLULAR FUNCTIONS AND PHENOTYPES OF IAP
- •3. IN VIVO FUNCTIONS OF IAP FAMILY PROTEINS
- •4. SUBCELLULAR LOCATIONS OF IAP
- •8. IAP–IAP INTERACTIONS
- •10. ENDOGENOUS ANTAGONISTS OF IAP
- •11. IAPs AND DISEASE
- •SUGGESTED READINGS
- •1. INTRODUCTION
- •2.1. The CD95 (Fas/APO-1) system
- •2.1.1. CD95 and CD95L: discovery of the first direct apoptosis-inducing receptor-ligand system
- •2.1.2. Biochemistry of CD95 apoptosis signaling
- •2.2. The TRAIL (Apo2L) system
- •3.1. The TNF system
- •3.1.1. Biochemistry of TNF signal transduction
- •3.1.2. TNF and TNF blockers in the clinic
- •3.2. The DR3 system
- •4. THE DR6 SYSTEM
- •6. CONCLUDING REMARKS AND OUTLOOK
- •SUGGESTED READINGS
- •4 Mitochondria and Cell Death
- •1. INTRODUCTION
- •2. MITOCHONDRIAL PHYSIOLOGY
- •3. THE MITOCHONDRIAL PATHWAY OF APOPTOSIS
- •9. CONCLUSIONS
- •SUGGESTED READINGS
- •1. INTRODUCTION
- •3. INHIBITING APOPTOSIS
- •4. INHIBITING THE INHIBITORS
- •6. THE BCL-2 FAMILY AND CANCER
- •SUGGESTED READINGS
- •6 Endoplasmic Reticulum Stress Response in Cell Death and Cell Survival
- •1. INTRODUCTION
- •2. THE ESR IN YEAST
- •3. THE ESR IN MAMMALS
- •4. THE ESR AND CELL DEATH
- •5. THE ESR IN DEVELOPMENT AND TISSUE HOMEOSTASIS
- •6. THE ESR IN HUMAN DISEASE
- •7. CONCLUSION
- •7 Autophagy – The Liaison between the Lysosomal System and Cell Death
- •1. INTRODUCTION
- •2. AUTOPHAGY
- •2.2. Physiologic functions of autophagy
- •2.3. Autophagy and human pathology
- •3. AUTOPHAGY AND CELL DEATH
- •3.1. Autophagy as anti–cell death mechanism
- •3.2. Autophagy as a cell death mechanism
- •3.3. Molecular players of the autophagy–cell death cross-talk
- •4. AUTOPHAGY, CELLULAR DEATH, AND CANCER
- •5. CONCLUDING REMARKS AND PENDING QUESTIONS
- •SUGGESTED READINGS
- •8 Cell Death in Response to Genotoxic Stress and DNA Damage
- •1. TYPES OF DNA DAMAGE AND REPAIR SYSTEMS
- •2. DNA DAMAGE RESPONSE
- •2.2. Transducers
- •2.3. Effectors
- •4. CHROMATIN MODIFICATIONS
- •5. CELL CYCLE CHECKPOINT REGULATION
- •6. WHEN REPAIR FAILS: SENESCENCE VERSUS APOPTOSIS
- •6.1. DNA damage response and the induction of apoptosis
- •6.2. p53-independent mechanisms of apoptosis
- •6.3. DNA damage response and senescence induction
- •7. DNA DAMAGE FROM OXIDATIVE STRESS
- •SUGGESTED READINGS
- •9 Ceramide and Lipid Mediators in Apoptosis
- •1. INTRODUCTION
- •3.1. Basic cell signaling often involves small molecules
- •3.2. Sphingolipids are cell-signaling molecules
- •3.2.1. Ceramide induces apoptosis
- •3.2.2. Ceramide accumulates during programmed cell death
- •3.2.3. Inhibition of ceramide production alters cell death signaling
- •4.1. Ceramide is generated through SM hydrolysis
- •4.3. aSMase can be activated independently of extracellular receptors to regulate apoptosis
- •4.4. Controversial aspects of the role of aSMase in apoptosis
- •4.5. De novo ceramide synthesis regulates programmed cell death
- •4.6. p53 and Bcl-2–like proteins are connected to de novo ceramide synthesis
- •4.7. The role and regulation of de novo synthesis in ceramide-mediated cell death is poorly understood
- •5. CONCLUDING REMARKS AND FUTURE DIRECTIONS
- •5.1. Who? (Which enzyme?)
- •5.2. What? (Which ceramide?)
- •5.3. Where? (Which compartment?)
- •5.4. When? (At what steps?)
- •5.5. How? (Through what mechanisms?)
- •5.6. What purpose?
- •6. SUMMARY
- •SUGGESTED READINGS
- •1. General Introduction
- •1.1. Cytotoxic lymphocytes and apoptosis
- •2. CYTOTOXIC GRANULES AND GRANULE EXOCYTOSIS
- •2.1. Synthesis and loading of the cytotoxic granule proteins into the secretory granules
- •2.2. The immunological synapse
- •2.3. Secretion of granule proteins
- •2.4. Uptake of proapoptotic proteins into the target cell
- •2.5. Activation of death pathways by granzymes
- •3. GRANULE-BOUND CYTOTOXIC PROTEINS
- •3.1. Perforin
- •3.2. Granulysin
- •3.3. Granzymes
- •3.3.1. GrB-mediated apoptosis
- •3.3.2. GrA-mediated cell death
- •3.3.3. Orphan granzyme-mediated cell death
- •5. CONCLUSIONS
- •REFERENCES
- •Part II Cell Death in Tissues and Organs
- •1.1. Death by trophic factor deprivation
- •1.2. Key molecules regulating neuronal apoptosis during development
- •1.2.1. Roles of caspases and Apaf-1 in neuronal cell death
- •1.2.2. Role of Bcl-2 family members in neuronal cell death
- •1.3. Signal transduction from neurotrophins and neurotrophin receptors
- •1.3.1. Signals for survival
- •1.3.2. Signals for death
- •2.1. Apoptosis in neurodegenerative diseases
- •2.1.4. Amyotrophic lateral sclerosis
- •2.2. Necrotic cell death in neurodegenerative diseases
- •2.2.1. Calpains
- •2.2.2. Cathepsins
- •3. CONCLUSIONS
- •ACKNOWLEDGMENT
- •SUGGESTED READINGS
- •ACKNOWLEDGMENT
- •SUGGESTED READINGS
- •1. INTRODUCTION
- •5. S-NITROSYLATION OF PARKIN
- •7. POTENTIAL TREATMENT OF EXCESSIVE NMDA-INDUCED Ca2+ INFLUX AND FREE RADICAL GENERATION
- •8. FUTURE THERAPEUTICS: NITROMEMANTINES
- •9. CONCLUSIONS
- •Acknowledgments
- •SUGGESTED READINGS
- •3. MITOCHONDRIAL PERMEABILITY TRANSITION ACTIVATED BY Ca2+ AND OXIDATIVE STRESS
- •4.1. Mitochondrial apoptotic pathways
- •4.2. Bcl-2 family proteins
- •4.3. Caspase-dependent apoptosis
- •4.4. Caspase-independent apoptosis
- •4.5. Calpains in ischemic neural cell death
- •5. SUMMARY
- •ACKNOWLEDGMENTS
- •SUGGESTED READINGS
- •1. INTRODUCTION
- •2. HISTORICAL ANTECEDENTS
- •7.1. Activation of p21 waf1/cip1: Targeting extrinsic and intrinsic pathways to death
- •8. CONCLUSION
- •ACKNOWLEDGMENTS
- •REFERENCES
- •16 Apoptosis and Homeostasis in the Eye
- •1.1. Lens
- •1.2. Retina
- •2. ROLE OF APOPTOSIS IN DISEASES OF THE EYE
- •2.1. Glaucoma
- •2.2. Age-related macular degeneration
- •4. APOPTOSIS AND OCULAR IMMUNE PRIVILEGE
- •5. CONCLUSIONS
- •SUGGESTED READINGS
- •17 Cell Death in the Inner Ear
- •3. THE COCHLEA IS THE HEARING ORGAN
- •3.1. Ototoxic hair cell death
- •3.2. Aminoglycoside-induced hair cell death
- •3.3. Cisplatin-induced hair cell death
- •3.4. Therapeutic strategies to prevent hair cell death
- •3.5. Challenges to studies of hair cell death
- •4. SPIRAL GANGLION NEURON DEATH
- •4.1. Neurotrophic support from sensory hair cells and supporting cells
- •4.2. Afferent activity from hair cells
- •4.3. Molecular manifestations of spiral ganglion neuron death
- •4.4. Therapeutic interventions to prevent SGN death
- •ACKNOWLEDGMENTS
- •SUGGESTED READINGS
- •18 Cell Death in the Olfactory System
- •1. Introduction
- •2. Anatomical Aspects
- •3. Life and Death in the Olfactory System
- •3.1. Olfactory epithelium
- •3.2. Olfactory bulb
- •REFERENCES
- •1. Introduction
- •3.1. Beta cell death in the development of T1D
- •3.2. Mechanisms of beta cell death in type 1 diabetes
- •3.2.1. Apoptosis signaling pathways downstream of death receptors and inflammatory cytokines
- •3.2.2. Oxidative stress
- •3.3. Mechanisms of beta cell death in type 2 diabetes
- •3.3.1. Glucolipitoxicity
- •3.3.2. Endoplasmic reticulum stress
- •5. SUMMARY
- •Acknowledgments
- •REFERENCES
- •20 Apoptosis in the Physiology and Diseases of the Respiratory Tract
- •1. APOPTOSIS IN LUNG DEVELOPMENT
- •2. APOPTOSIS IN LUNG PATHOPHYSIOLOGY
- •2.1. Apoptosis in pulmonary inflammation
- •2.2. Apoptosis in acute lung injury
- •2.3. Apoptosis in chronic obstructive pulmonary disease
- •2.4. Apoptosis in interstitial lung diseases
- •2.5. Apoptosis in pulmonary arterial hypertension
- •2.6. Apoptosis in lung cancer
- •SUGGESTED READINGS
- •21 Regulation of Cell Death in the Gastrointestinal Tract
- •1. INTRODUCTION
- •2. ESOPHAGUS
- •3. STOMACH
- •4. SMALL AND LARGE INTESTINE
- •5. LIVER
- •6. PANCREAS
- •7. SUMMARY AND CONCLUDING REMARKS
- •SUGGESTED READINGS
- •22 Apoptosis in the Kidney
- •1. NORMAL KIDNEY STRUCTURE AND FUNCTION
- •3. APOPTOSIS IN ADULT KIDNEY DISEASE
- •4. REGULATION OF APOPTOSIS IN KIDNEY CELLS
- •4.1. Survival factors
- •4.2. Lethal factors
- •4.2.1. TNF superfamily cytokines
- •4.2.2. Other cytokines
- •4.2.3. Glucose
- •4.2.4. Drugs and xenobiotics
- •4.2.5. Ischemia-reperfusion and sepsis
- •5. THERAPEUTIC APPROACHES
- •SUGGESTED READINGS
- •1. INTRODUCTION
- •2. APOPTOSIS IN THE NORMAL BREAST
- •2.1. Occurrence and role of apoptosis in the developing breast
- •2.2.2. Death ligands and death receptor pathway
- •2.2.4. LIF-STAT3 proapoptotic signaling
- •2.2.5. IGF survival signaling
- •2.2.6. Regulation by adhesion
- •2.2.7. PI3K/AKT pathway: molecular hub for survival signals
- •2.2.8. Downstream regulators of apoptosis: the BCL-2 family members
- •3. APOPTOSIS IN BREAST CANCER
- •3.1. Apoptosis in breast tumorigenesis and cancer progression
- •3.2. Molecular dysregulation of apoptosis in breast cancer
- •3.2.1. Altered expression of death ligands and their receptors in breast cancer
- •3.2.2. Deregulation of prosurvival growth factors and their receptors
- •3.2.3. Alterations in cell adhesion and resistance to anoikis
- •3.2.4. Enhanced activation of the PI3K/AKT pathway in breast cancer
- •3.2.5. p53 inactivation in breast cancer
- •3.2.6. Altered expression of BCL-2 family of proteins in breast cancer
- •5. CONCLUSION
- •SUGGESTED READINGS
- •1. INTRODUCTION
- •2. DETECTING CELL DEATH IN THE FEMALE GONADS
- •4. APOPTOSIS AND FEMALE REPRODUCTIVE AGING
- •6. CONCLUDING REMARKS
- •REFERENCES
- •25 Apoptotic Signaling in Male Germ Cells
- •1. INTRODUCTION
- •3.1. Murine models
- •3.2. Primate models
- •3.3. Pathways of caspase activation and apoptosis
- •3.4. Apoptotic signaling in male germ cells
- •5. P38 MITOGEN-ACTIVATED PROTEIN KINASE (MAPK) AND NITRIC OXIDE (NO)–MEDIATED INTRINSIC PATHWAY SIGNALING CONSTITUTES A CRITICAL COMPONENT OF APOPTOTIC SIGNALING IN MALE GERM CELLS AFTER HORMONE DEPRIVATION
- •11. CONCLUSIONS AND PERSPECTIVES
- •REFERENCES
- •26 Cell Death in the Cardiovascular System
- •1. INTRODUCTION
- •2. CELL DEATH IN THE VASCULATURE
- •2.1. Apoptosis in the developing blood vessels
- •2.2. Apoptosis in atherosclerosis
- •2.2.1. Vascular smooth muscle cells
- •2.2.2. Macrophages
- •2.2.3. Regulation of apoptosis in atherosclerosis
- •2.2.4. Necrosis and autophagy in atherosclerosis
- •3. CELL DEATH IN THE MYOCARDIUM
- •3.1. Cell death in myocardial infarction
- •3.1.1. Apoptosis in myocardial infarction
- •3.1.2. Necrosis in myocardial infarction
- •3.1.3. Autophagy in myocardial infarction
- •3.2. Cell death in heart failure
- •3.2.1. Apoptosis in heart failure
- •3.2.2. Necrosis in heart failure
- •3.2.3. Autophagy in heart failure
- •4. CONCLUDING REMARKS
- •ACKNOWLEDGMENTS
- •REFERENCES
- •27 Cell Death Regulation in Muscle
- •1. INTRODUCTION TO MUSCLE
- •1.1. Skeletal muscle adaptation to endurance training
- •1.2. Myonuclear domains
- •2. MITOCHONDRIALLY MEDIATED APOPTOSIS IN MUSCLE
- •2.1. Skeletal muscle apoptotic susceptibility
- •4. APOPTOSIS IN MUSCLE DURING AGING AND DISEASE
- •4.1. Aging
- •4.2. Type 2 diabetes mellitus
- •4.3. Cancer cachexia
- •4.4. Chronic heart failure
- •6. CONCLUSION
- •SUGGESTED READINGS
- •28 Cell Death in the Skin
- •1. INTRODUCTION
- •2. CELL DEATH IN SKIN HOMEOSTASIS
- •2.1. Cornification and apoptosis
- •2.2. Death receptors in the skin
- •3. CELL DEATH IN SKIN PATHOLOGY
- •3.1. Sunburn
- •3.2. Skin cancer
- •3.3. Necrolysis
- •3.4. Pemphigus
- •3.5. Eczema
- •3.6. Graft-versus-host disease
- •4. CONCLUDING REMARKS AND PERSPECTIVES
- •ACKNOWLEDGMENTS
- •SUGGESTED READINGS
- •29 Apoptosis and Cell Survival in the Immune System
- •2.1. Survival of early hematopoietic progenitors
- •2.2. Sizing of the T-cell population
- •2.2.1. Establishing central tolerance
- •2.2.2. Peripheral tolerance
- •2.2.3. Memory T cells
- •2.3. Control of apoptosis in B-cell development
- •2.3.1. Early B-cell development
- •2.3.2. Deletion of autoreactive B cells
- •2.3.3. Survival and death of activated B cells
- •3. IMPAIRED APOPTOSIS AND LEUKEMOGENESIS
- •4. CONCLUSIONS
- •ACKNOWLEDGMENTS
- •REFERENCES
- •30 Cell Death Regulation in the Hematopoietic System
- •1. INTRODUCTION
- •2. HEMATOPOIETIC STEM CELLS
- •4. ERYTHROPOIESIS
- •5. MEGAKARYOPOIESIS
- •6. GRANULOPOIESIS
- •7. MONOPOIESIS
- •8. CONCLUSION
- •ACKNOWLEDGMENTS
- •REFERENCES
- •31 Apoptotic Cell Death in Sepsis
- •1. INTRODUCTION
- •2. HOST INFLAMMATORY RESPONSE TO SEPSIS
- •3. CLINICAL OBSERVATIONS OF CELL DEATH IN SEPSIS
- •3.1. Sepsis-induced apoptosis
- •3.2. Necrotic cell death in sepsis
- •4.1. Central role of apoptosis in sepsis mortality: immune effector cells and gut epithelium
- •4.2. Apoptotic pathways in sepsis-induced immune cell death
- •4.3. Investigations implicating the extrinsic apoptotic pathway in sepsis
- •4.4. Investigations implicating the intrinsic apoptotic pathway in sepsis
- •5. THE EFFECT OF APOPTOSIS ON THE IMMUNE SYSTEM
- •5.1. Cellular effects of an increased apoptotic burdens
- •5.2. Network effects of selective loss of immune cell types
- •5.3. Studies of immunomodulation by apoptotic cells in other fields
- •7. CONCLUSION
- •REFERENCES
- •32 Host–Pathogen Interactions
- •1. INTRODUCTION
- •2. FROM THE PATHOGEN PERSPECTIVE
- •2.1. Commensals versus pathogens
- •2.2. Pathogen strategies to infect the host
- •3. HOST DEFENSE
- •3.1. Antimicrobial peptides
- •3.2. PRRs and inflammation
- •3.2.1. TLRs
- •3.2.2. NLRs
- •3.2.3. The Nod signalosome
- •3.2.4. The inflammasome
- •3.3. Cell death
- •3.3.1. Apoptosis and pathogen clearance
- •3.3.2. Pyroptosis
- •3.2.3. Caspase-independent cell death
- •3.2.4. Autophagy and autophagic cell death
- •4. CONCLUSIONS
- •REFERENCES
- •Part III Cell Death in Nonmammalian Organisms
- •1. PHENOTYPE AND ASSAYS OF YEAST APOPTOSIS
- •2.1. Pheromone-induced cell death
- •2.1.1. Colony growth
- •2.1.2. Killer-induced cell death
- •3. EXTERNAL STIMULI THAT INDUCE APOPTOSIS IN YEAST
- •4. THE GENETICS OF YEAST APOPTOSIS
- •5. PROGRAMMED AND ALTRUISTIC AGING
- •SUGGESTED READINGS
- •34 Caenorhabditis elegans and Apoptosis
- •1. Overview
- •2. KILLING
- •3. SPECIFICATION
- •4. EXECUTION
- •4.1. DNA degradation
- •4.2. Mitochondrial elimination
- •4.3. Engulfment
- •5. SUMMARY
- •SUGGESTED READINGS
- •35 Apoptotic Cell Death in Drosophila
- •2. DROSOPHILA CASPASES AND PROXIMAL REGULATORS
- •6. CLOSING COMMENTS
- •SUGGESTED READINGS
- •36 Analysis of Cell Death in Zebrafish
- •1. INTRODUCTION
- •2. WHY USE ZEBRAFISH TO STUDY CELL DEATH?
- •2.2. Molecular techniques to rapidly assess gene function in embryos
- •2.2.1. Studies of gene function using microinjections into early embryos
- •2.2.2. In situ hybridization and immunohistochemistry
- •2.3. Forward genetic screening
- •2.4. Drug and small-molecule screening
- •2.5. Transgenesis
- •2.6. Targeted knockouts
- •3.1. Intrinsic apoptosis
- •3.2. Extrinsic apoptosis
- •3.3. Chk-1 suppressed apoptosis
- •3.4. Anoikis
- •3.5. Autophagy
- •3.6. Necrosis
- •4. DEVELOPMENTAL CELL DEATH IN ZEBRAFISH EMBRYOS
- •5. THE P53 PATHWAY
- •6. PERSPECTIVES AND FUTURE DIRECTIONS
- •SUGGESTED READING
96 |
THOMAS D. MULLEN, RUSSELL W. JENKINS, LINA M. OBEID, AND YUSUF A. HANNUN |
death was preceded by ceramide accumulation that was not seen with genetic or pharmacological depletion of aSMase. Additionally, administration of exogenous aSMase restores the cell death in aSMase-deficient hepatocytes in response to TNF-α. Adenoviral-mediated expression in the liver of the cDNA of neutral ceramidase (nCDase), an enzyme that metabolizes ceramide to sphingosine, protected mice from TNF-α–induced elevations in ceramide and liver damage. Over-expression of nCDase also resulted in increased activation of the PI3K/Akt prosurvival pathway, presumably shunting sphingosine through sphingosine kinase and resulting in enhanced levels of the prosurvival sphingolipid S1P. Similar studies have demonstrated a capacity of ceramidase and sphingosine kinase to modulate cell fate, leading some researchers to use the term rheostat to describe a hypothetical regulatory mechanism that specifically balances levels of proapoptotic ceramide and antiapoptotic S1P.
Another cell surface receptor of the TNF family whose activation is more commonly associated with cell death is CD95 (also known as Fas/Apo-1/TNFR6). The role of aSMase in CD95 signaling has centered around the observation that capping or clustering of CD95 is defective in the absence of a functional aSMase. The first report of in vivo significance of the aSMase/ceramide pathway in CD95 ligand (CD95L)-induced cell death demonstrated that aSMase knockout mice were protected from hepatocyte apoptosis after CD95 activation in a model of autoimmune hepatitis. Requirement for ceramide has been further supported by studies using exogenous ceramide to reverse CD95resistance in aSMase-null mice. Exogenous ceramide bypasses the metabolic block resulting from genetic absence of aSMase, demonstrating a strict requirement for ceramide in receptor-mediated cell death.
4.3. aSMase can be activated independently of extracellular receptors to regulate apoptosis
In addition to its role in death receptor–mediated cell death, the aSMase/ceramide pathway is activated by several toxic stimuli (e.g., radiation, chemotherapeutic drugs, toxic metals) that lead to ceramide-dependent cell death through cellular mechanisms that are largely unknown (see Clinical Case 9-1: aSMase and Wilson’s disease). The earliest connection between defects in sphingomyelinase-mediated ceramide generation and diminished apoptosis came in 1996 when the Kolesnick lab demonstrated an in vitro and an in vivo requirement for a functional aSMase in ionizing radiation (IR)– induced apoptosis (Santana et al. 1996). IR induces
apoptosis by producing double-strand DNA breaks and can signal cell death through a p53-dependent pathway. However, human lymphoblasts from NPD patients lacking aSMase exhibit resistance to IR, and sensitivity can be restored by retroviral delivery of aSMase cDNA. In vivo protection from IR-induced apoptosis in aSMasedeficient mice is also seen in multiple tissues after whole-body irradiation; these include the lung, central nervous system, intestinal tract, and spleen. The authors also compared the apoptotic response in aSMase knockout mice with mice lacking p53. The intriguing results are that although certain tissues seem to rely on aSMase as a primary mediator of IR-induced apoptosis (e.g., microvascular endothelial cells), other tissues seem to have a lesser requirement for aSMase and a greater need for functional p53 (e.g., thymus). Thus the role and/or the regulation of aSMase may be unique to cell types that are less reliant on p53 signaling.
Besides its function in radiation-induced death of normal tissues, aSMase may represent an important mediator of radiation-induced death of cancer cells. Given the utility of IR in the treatment of various cancers, the role of aSMase in mediating IR-induced tumor regression has also been examined. IR-induced tumor regression was shown to require intact aSMase, and the effect was reported to involve IR-induced microvascular endothelial cell apoptosis. Garcia-Barros et al. (2003) demonstrated that cancer cells grew to larger overall tumor size implanted subcutaneously in aSMase-null mice as compared with wild-type mice, but more importantly, in response to IR treatment, the fibrosarcoma cells grown in aSMase-null mice did not respond, whereas tumor size was diminished in the wild-type mice. This effect was later confirmed to involve endothelial cell viability of the host mouse, although the role of primary endothelial injury in IR-induced tumor cell death remains a highly contentious issue.
4.4. Controversial aspects of the role of aSMase in apoptosis
Although these examples may be compelling, a universal role for the aSMase/ceramide pathway in cell death cannot be fully substantiated by the available evidence. For example, aSMase deficiency does not confer protection from cell death in all tissues to the same extent. Lin et al. (2000) reported that CD95 activation induced apoptosis in wild-type and SMPD1–/– B and T lymphocytes equally well, whereas hepatocytes from SMPD1–/– mice were markedly protected from death. In addition to differences in cell type dependence, it has been shown that protection from cell death in mouse
CERAMIDE AND LIPID MEDIATORS IN APOPTOSIS |
97 |
|
|
CLINICAL CASE STUDY 9-1: THE aSMASE/CERAMIDE |
tocytes, Cu2+ induced activation of aSMase in leukocyte cel- |
PATHWAY IN WILSON’S DISEASE |
lular extracts, but importantly, Cu2+ also stimulated secre- |
|
tion of aSMase into the culture medium. Incubation of |
Wilson’s disease (WD) is an autosomal-recessive metabolic |
purified erythrocytes with conditioned media from Cu2+- |
disorder resulting from mutations in ATP7B, a gene that |
treated leukocytes induced ceramide accumulation in ery- |
encodes a P-type ATPase Cu2+ transporter. Defects in ATB7B |
throcytes. The increases in ceramide were associated with |
lead to dysregulation of Cu2+ homeostasis, ultimately caus- |
PS externalization, a feature of programmed cell death in |
ing build-up of copper in tissues throughout the body. Over |
erythrocytes (also known as eryptosis). More importantly, |
time the accumulation of copper can reach toxic levels in |
conditioned media produced by leukocytes from aSMase |
many tissues, including the liver, skeletal muscle, bone, ner- |
knockout mice did not induce PS exposure and eryptosis |
vous system, and formed elements of blood. Excessive lev- |
in response to Cu2+ . These data support a model in which |
els of Cu2+ in the liver and blood result in death of hep- |
secretory aSMase is released by Cu2+-overloaded leukocytes |
atocytes and erythrocytes, respectively, and consequently |
to generate ceramide on erythrocytes – ultimately leading |
produce hepatitis and anemia. |
to erythrocyte loss and anemia. |
Lang et al. (2007) recently provided evidence for involve- |
Although the upstream mechanisms of Cu2+-induced |
ment of the aSMase/ceramide pathway in the induction of |
activation of aSMase remain unknown, the downstream |
apoptosis in response to toxic levels of Cu2+. In primary hep- |
role of aSMase-derived ceramide in hepatocellular death |
atocytes, Cu2+ overload induced a dose-dependent increase |
and anemia supports a crucial role for ceramide in cell |
in total ceramide that was mirrored by elevations in aSMase |
death signaling and identifies aSMase as a novel thera- |
activity. Genetic or pharmacological loss of aSMase pro- |
peutic target in the treatment of WD. ATP7B-defecient rats |
tected hepatocytes from Cu2+ intoxication and cell death, |
treated with the aSMase inhibitor amitryptyline experience |
demonstrating that aSMase is an important mediator of |
reduced aSMase activity, liver fibrosis, and eryptosis. More |
Cu2+ toxicity. |
significantly, aSMase inhibition confers a survival advantage |
The authors also investigated the role of the aSMase/ |
to these rats compared with untreated controls. Although |
ceramide pathway in the pathophysiology of Cu2+-induced |
still in its infancy, modulation of aSMase activity may one |
anemia of WD. Patients with WD have elevated aSMase |
day be a viable therapeutic strategy in the treatment |
activity in plasma derived from leukocytes. Similarly to hepa- |
of WD. |
|
|
embryonic fibroblasts (MEFs) from SMPD1–/– mice is stress-type specific. Lozano et al. (2001) demonstrated that SMPD1–/– MEFs were markedly protected from IR but not from staurosporine induced cell death. These MEFs also exhibited intermediate protection from other forms of cell death (e.g., serum withdrawal and TNF-α). Overall, the evidence suggests that the aSMase/ceramide pathway may be “recruited” only in certain forms of cell death. Moreover, even when using a variety of aSMase-deficient Niemann-Pick cell lines, Bezombes et al. (2001) demonstrated that aSMase was dispensable for apoptosis in response to established “inducers” of aSMase, including IR and CD95L. Nix and Stoffel (2000) showed that aSMase-deficient T cells were actually more susceptible to CD95L stimulation, further highlighting the varied nature of responses that have been reported. Thus, although some of these discrepant findings can be attributed to different cell lines or stimuli, clearly they cannot account for variability within specific model systems. Given the conflicting reports, the aSMase/ceramide pathway may not represent an integral component of the cell death
program, but rather a modifier of apoptotic signaling that is preferentially recruited under certain conditions. Future experimentation using gain-of-function experiments to complement loss-of-function studies, as well as the development of tools (e.g., specific inhibitors) to identify the form(s) of aSMase responsible for generating plasma membrane ceramide (i.e., secretory vs. lysosomal aSMase), will help to ascertain the signaling function of the aSMase/ceramide pathway.
4.5. De novo ceramide synthesis regulates programmed cell death
As mentioned above, SMases and SMase-dependent ceramide production can control programmed cell death in a variety of contexts. However, as research into sphingolipid signaling expanded in the 1990s, it became apparent that there are SMase-independent pathways of ceramide accumulation during cell death. Researchers discovered death-induced ceramide production in the absence of increased SM hydrolysis. Furthermore, the discovery of small-molecule inhibitors of de novo
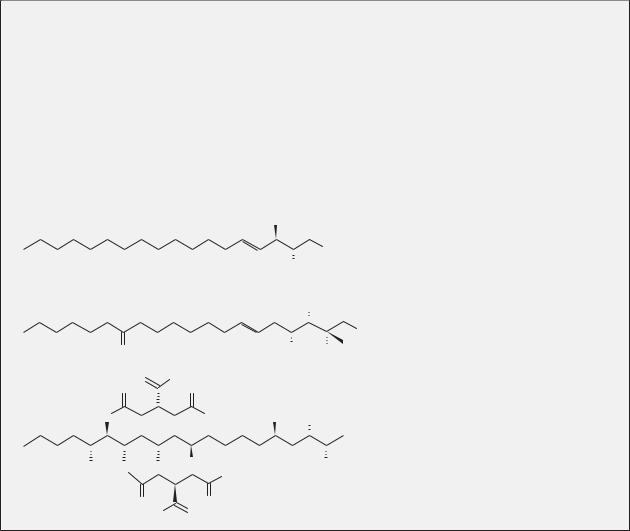
98 |
THOMAS D. MULLEN, RUSSELL W. JENKINS, LINA M. OBEID, AND YUSUF A. HANNUN |
Box 9-3. Fungal metabolites are essential tools for the study of sphingolipids
Two major fungal metabolites have become standard tools for the sphingolipid researcher. The first is myriocin, also known as ISP-1, a fungal metabolite of Isaria sinclairii that was discovered to have immunosuppressive e ects. The structure of myriocin is highly similar to that of sphingosine (Figure B9-3B), leading researchers to find that the compound is a potent inhibitor of serine palmitoyltransferase. Since its discovery, myriocin has been used extensively to characterize sphingolipid biochemistry and sphingolipid-mediated biology. The other inhibitor that has enjoyed widespread use is fumonisin B1 (FB1), a member of a family of fungal toxins called fumonisins that are produced by Fusarium moniliforme, a fungus that infects several grain species, especially corn (maize). Like myriocin, the structure of fumonisin bears resemblance to that of sphingosine (Figure B9-3C) and was therefore tested for its e ects on sphingolipid metabolism. FB1 inhibits the acylation of dihydrosphingosine and sphingosine by CerS, thus reducing ceramide production and causing the accumulation of sphingoid bases. As with myriocin, FB1 has been invaluable to the sphingolipid researcher and essential to the study of de novo ceramide synthesis.
A
B
C
OH
OH
NH2
OH
OH
O |
|
OH |
NH2 COOH |
O |
O H |
|
|
O |
O |
|
|
O |
OH |
OH |
OH |
O |
OH |
|
NH2 |
|
OH |
||
|
|
|
|
O |
O |
|
|
HO |
O |
|
|
Figure B9-3. Structures of sphingosine and sphi- ngolipid-like fungal products. (A) Sphingosine, (B) myriocin, and (C) fumonisin B1.
sphingolipid synthesis allowed investigators to use these compounds to probe the mechanisms of ceramidemediated apoptosis (Box 9-3).
De novo synthesis as a means of death-induced ceramide generation was first described for the chemotherapeutic agent daunorubicin in 1995. Researchers found increasing ceramide generation over the course of hours after treatment of leukemia cells with daunorubicin. The accumulation of ceramide occurred in the absence of SMase activation. Instead, CerS activity was increased, and the CerS inhibitor FB1 was able to inhibit ceramide production and cell death. Since these preliminary studies, a myriad of proapoptotic agents have been shown to induce de novo ceramide generation in several model systems (Figure 9-7).
One system that illustrates a unique requirement for CerS and de novo ceramide synthesis is the induction of B-cell apoptosis through activation of the B-cell receptor (BcR). Treatment of B cells with anti–immunoglobulin M
causes BcR activation, ceramide production, mitochondrial dysfunction, and apoptosis. Treatment with FB1 prevents the production of ceramide and downstream apoptotic events. Additional studies show that de novo ceramide generation participates in BcR-mediated proteasome activation and degradation of the endogenous caspase inhibitor X-linked inhibitor of apoptosis (XIAP). It is not known how ceramide directly controls these downstream effects, but the identification of targets for endogenously generated ceramide is currently an important goal in this field of research.
What enzymes are involved in de novo ceramide production during cell death? The answer to this question depends on the combination of death inducer and model system. Some studies have shown deathinduced activation of SPT, whereas others provide evidence for ceramide synthase activation. For example, cannabinoids such as 9-tetrahydrocannabinol induce cell death in glioma cells that are dependent on
CERAMIDE AND LIPID MEDIATORS IN APOPTOSIS |
99 |
activation of SPT. On the other hand, irradiation of human keratinocytes with UVB light activates CerS to produce ceramide. In many cases, ceramide accumulation is shown to be dependent on de novo synthesis (via pharmacological inhibition), but whether activation of ceramide synthesis per se occurs at the enzymatic level has yet to be demonstrated for the majority of cases and demands further investigation.
4.6. p53 and Bcl-2–like proteins are connected to de novo ceramide synthesis
Despite being studied in numerous cell death models, the mechanisms of death-induced de novo ceramide generation, either by SPT or ceramide synthases, are poorly defined. One key upstream mediator that has been identified is the tumor suppressor p53, which accumulates in response to DNA damage. Treatment of Molt-4 leukemia cells with the transcriptional inhibitor dactinomycin or γ-irradiation causes p53 accumulation, ceramide production, and apoptosis. Blocking p53 by expressing the human papillomavirus protein E6 – an inhibitor of p53 – prevents ceramide accumulation and cell death. Other clues about the regulation of de novo ceramide synthesis come from studies of the role of the DNA damage response kinase ATM in regulating IRinduced intestinal damage. Loss of function ATM sensitizes mice to low-dose radiation and promotes ceramide synthase activation. How p53 connects the genotoxic stress response to the modulation of ceramide levels remains to be answered. However, recent studies begin to implicate sphingosine kinase (SK1) as a possible mediator of the p53 effects. Activation of p53 was shown to result in loss of SK1, possibly through proteolysis with subsequent accumulation of ceramide.
Some studies show that exogenous ceramide can promote the increase of p53 itself, resulting in the activation of a p53-dependent cell death pathway. On the other hand, exogenous ceramide can also promote cell death in the absence of functional p53. The latter evidence is exciting from a therapeutic standpoint. Unlike DNAdamaging chemotherapeutics, ceramide or ceramide analogs may be effective therapeutic adjuncts in the treatment of cancers lacking functional p53.
Members of the Bcl-2 family of proteins are critical regulators of apoptosis. Generally, they are the gatekeepers to the mitochondrial pathway of programmed cell death – a critical step in the commitment of a cell to its own demise. Bcl-2 family members, including both proapoptotic and antiapoptotic proteins, can also control late ceramide production in ways that are not fully understood. In
some cell systems, late ceramide production occurs upstream of Bcl-2 action and the mitochondrial pathway, whereas in others ceramide production occurs downstream of these events. In MCF-7 breast adenocarcinoma cells, over-expression of either Bcl-2 or Bcl-XL inhibits TNF-α or camptothecin-induced ceramide generation and cell death. However, in other systems (e.g., vincristine-induced death in acute lymphoblastic leukemia) ceramide generation is upstream of Bcl-2–like proteins. Along similar lines, ceramide may influence mitochondrial permeabilization, either through direct protein-lipid interactions with proapoptotic proteins such as Bax or by influencing the formation of channels in the mitochondrial outer membrane.
Several targets of de novo–generated ceramide have been described. De novo ceramide production has been linked to activation of the proteasome and degradation of the caspase inhibitor XIAP. In other studies, ceramide production was shown to specifically regulate splicing of Bcl-X and caspase-9; ceramide activates PP1 to promote dephosphorylation of the SR proteins, which regulate alternative splicing. De novo ceramide generation leads to the production of proapoptotic variants of Bcl-X and caspase-9 (Bcl-XS and caspase-9b, respectively).
Another system that illustrates de novo ceramidemediated death is apoptosis induced by cannabinoids. Cannabinoids induce apoptosis via binding extracellular receptors and promoting ceramide accumulation via the de novo pathway. In pancreatic cancer and glioma cells, the effects of ceramide appear to be mediated through the stress-associated protein p8. Although the mechanisms are still unclear, p8 connects ceramide to the ER stress-induced apoptosis via upregulation of the genes ATF-4 and TRB3. Despite these clues into the actions of ceramide, a detailed knowledge of effectors of de novo ceramide-mediated death remains elusive.
4.7. The role and regulation of de novo synthesis in ceramide-mediated cell death is poorly understood
Because of the conflicting evidence, it is currently impossible to make broad generalizations about late ceramide generation and its specific role in the regulation of proapoptotic signaling. Elucidating the detailed mechanisms of late ceramide generation – whether by de novo synthesis or not – remains an area of active research. Many studies have indicated that de novo synthesis is necessary for ceramide accumulation, but few have demonstrated bona fide activation of the enzymes of ceramide anabolism. Along similar lines, it is not known whether the degradative enzymes play a regulated role in controlling ceramide accumulation; as mentioned
